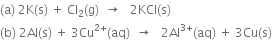
(a) The given reaction
В В В В В В В 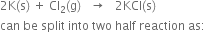
(i) Oxidation half-reaction.
В В В В В В В В В В В В В В В В 
(ii) Reduction half reaction:
В В В В В В В В В В В В В В В В 
(b) The given reaction
В В В В В В 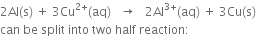
(i) Oxidation half reaction:
В В В В В В В В В В В 
(ii) Reduction half reaction:
В В В В В В В В 
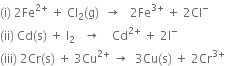
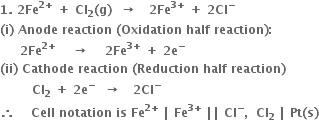
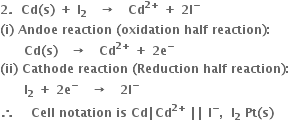
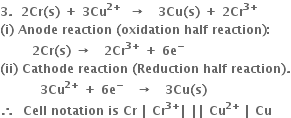




What is electrodeВ potential? Name the factors on which it depends.
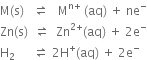
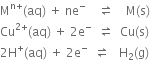
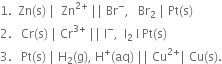
1. В For the cell
В В В 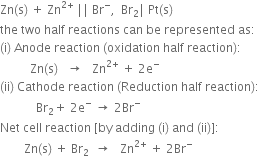
Positive terminal: В The bromine electrode where reduction takes place.
2. For the cellВ 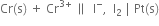
The two half reactions can be represented as:
(i) Anode reaction (oxidation half reaction):
В В В В В В В В В 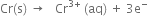
(ii) Cathode reaction (reduction half reaction):
В В В В В В В В 
Positive terminal. The iodine electrode where reduction takes place.
3. For cellВ 
the two half reactions can be represented as:
(i) Anode reaction (oxidation half reaction):
В В В В В В В В В В В 
(ii) Cathode reaction (reduction half reaction):
В В В В В В В В В В 
Net cell reaction: В It is obtained by adding equations (i) and (ii).
Positive terminal: The copper electrode where reduction takes place.
