 Multiple Choice Questions
Multiple Choice QuestionsOzonolysis of an organic compound gives formaldehyde as one of the products. This confirms the presence of,
two ethylenic double bonds
a vinyl group
an isopropyl group
an isopropyl group
Silver Mirror test is given by which one of the following compounds?
Acetaldehyde
Acetone
Formaldehyde
Formaldehyde
One mole of a symmetrical alkene on ozonolysis gives two moles of an aldehyde having a molecular mass of 44 u. The alkene is
Propene
1-butene
2-butene
2-butene
Which of the following, upon treatment with tert-BuONa followed by addition of bromine water, fails to decolourize the colour of bromine?
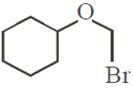
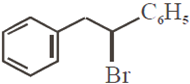
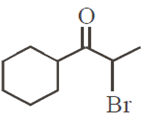

The correct sequence of reagents for the following conversion will be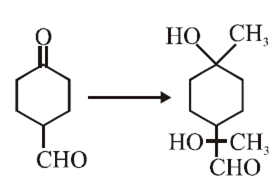
[Ag(NH3)2]+ OH–, H+/CH3OH, CH3MgBr
CH3MgBr, H+/CH3OH, [Ag(NH3)2]+ OH–
CH3MgBr, [Ag(NH3)2]+ OH–, H+/CH3OH
CH3MgBr, [Ag(NH3)2]+ OH–, H+/CH3OH
In Cannizzaro reaction given below ![]() the slowest step is
the slowest step is
The attack of ![]() at the carboxyl group
at the carboxyl group
The transfer of hydride to the carbonyl group
The abstraction of proton from the carboxylic group
The abstraction of proton from the carboxylic group
In the following sequence of reactions, the alkene affords the compound ‘B’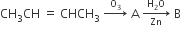
The compound B is
CH3CH2CHO
CH3COCH3
CH3CH2COCH3
CH3CH2COCH3
The compound formed as a result of oxidation of ethylbenzene by KMnO4 is
benzophenone
acetophenone
benzoic acid
benzoic acid
The increasing order of the rate of HCN addition to compounds A – D is
(A) HCHO
(B) CH3COCH3
(C) PhCOCH3
(D) PhCOPh
A < B < C < D
D < B < C < A
D < C < B < A
D < C < B < A
