 Short Answer Type
Short Answer TypeA boy weighing 40 kgf climbs up a stair of 30 steps each 20 cm high in 4 minutes and a girl weighing 30 kgf does the same in 3 minutes.
Compare:
(i) The work done by them.
(ii) The power developed by them
With reference to the terms Mechanical Advantage, Velocity Ratio and efficiency of a machine, name and define the term that will not change for machine of a given design.
Calculate the mass of ice required to lower the temperature of 300 g of water from 40o C to water at 0o C.
[Specific latent heat of ice = 336 J/g, specific heat capacity of water = 4.2 J/go C]
What do you understand by the following statements:
i) The heat capacity of the body is 60 JK-1
ii) The specific heat capacity of lead is 130 Jkg-1 K-1
A boy uses blue colour of light to find the refractive index o fglass. He then repeats the experiment using red colour of light. Will the refractive index be the same or different in the two cases? Give a reason to support your answer.
Copy the diagram given below and complete the path of the light ray till it emerges out of the prism. The critical angle of glass is 42o. In your diagram mark the angles wherever necessary.
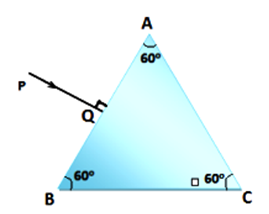
State the dependence of angle of deviation:
(i) On the refractive index of the material of the prism.
(ii) On the wavelength of light
The ratio of amplitude of two waves is 3:4. What is the ratio of their:
(i) loudness? (ii) Frequencies?
