 Multiple Choice Questions
Multiple Choice QuestionsDuring the process of digestion, the proteins present in food materials are hydrolysed in amino acids. The two enzymes involved in the process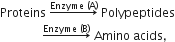
are respectively:
amylase and maltase
diastase and lipase
pepsin and trypsin
pepsin and trypsin
[NH(CH2)NHCO(CH2)4CO]n
co-polymer
addition polymer
thermosetting polymer
thermosetting polymer
A carbonyl compound reacts with hydrogen cyanide to form cyanohydrin which on hydrolysis forms a racemic mixture of alpha-hydroxy acid. The carbonyl compound is:
acetaldehyde
acetone
diethyl ketone
diethyl ketone
Which one of the following is a peptide hormone?
Glucagon
Testosterone
Thyroxin
Thyroxin
The major organic product in the reaction,
CH3 -O- CH(CH3)2 + HI → Product is:
CH3OH + (CH3)2 + CHI
ICH2OCH(CH3)2
CH3O CI(CH3)2
CH3O CI(CH3)2
A carbonyl compound reacts with hydrogen cyanide to form cyanohydrin which on hydrolysis forms a racemic mixture of alpha -hydroxy acid. The carbonyl compound is:
CH3-CH2-CH2COCH3
(CH3)2C=O
CH3CH2CHO
CH3CH2CHO
Self-condensation of two moles of ethyl acetate in presence of sodium ethoxide yields
ethyl butyrate
acetoacetic ester
methyl acetoacetate
methyl acetoacetate
Ethylene oxide when treated with Grignard reagent yields:
Secondary alcohol
tertiary alcohol
cyclopropyl alcohol
cyclopropyl alcohol
Which of the following is more basic than aniline?
Diphenylamine
Triphenylamine
p-nitroaniline
p-nitroaniline
D.
p-nitroaniline
Benzylamine C6H5 - NH2 is more basic than aniline because benzyl group C6H5CH2 is electron donating group due to +I effect. So, it is able to increase the electron density of N of -NH2 group. Thus due to higher electron density rate of donation of a free pair of electron is increased i.e, basic character is higher. Phenyl and nitro group are electron attractive groups. so they are able to decrease the electron density of N of NH2 group. Hence, they are less basic with aniline.
