 Multiple Choice Questions
Multiple Choice QuestionsIf it is assumed that decays only by emitting -and (-particles, the possible product of the decay is
Of the following compounds, the most acidic is
As2O3
P2O5
Sb2O3
Bi2O3
B.
P2O5
Generally, acidic nature of oxides increases along a period from left to right and decreases along a group from up to down.
Since As, P, Sb and Bi all belong to V group and arranged in the following manner :
P: As, Sb, Bi
:. P2O5 is the most and Bi2O3 is the least acidic.
The reaction that takes place when Cl2 gas is passed through conc. NaOH solution is
oxidation
reduction
displacement
disproportionation
Which of the following statements is not correct?
Silicon is extensively used as a semiconductor
Carborundum is SiC
Silicon occurs in free state in nature
Mica contains the element silicon
The addition of HBr to 2-pentene gives
2-bromopentane only
3-bromopentane only
2-bromopentane and 3-bromopentane
1-bromopentane and 3-bromopentane
ethylene can be separated from acetylene by passing the mixture through
fuming H2SO4
pyrogallol
ammoniacal Cu2Cl2
charcoal powder
In the compound the hybridisation of C-2 and C-3 carbons are respectively
sp3 and sp3
sp2 and sp3
sp2 and sp
sp3 and sp
The two structures written below represent
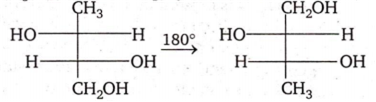
pair of diastereomers
pair of enantiomers
same molecule
both are optically inactive
