Multiple Choice Questions
Multiple Choice QuestionsHydrocarbon (A) reacts with bromine by substitution to form an alkyl bromide which by Wurtz reaction is converted to a gaseous hydrocarbon containing less than four carbon atoms. (A) is
CH ≡ CH
H2C = CH2
CH4
CH3-CH3
In the reaction,
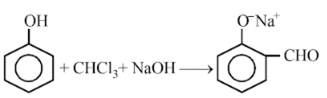
The electrophile involved is
Dichloromethyl cation (C+HCl2)
Formyl cation (C+HO)
Dichlorocarbene (:CCl2)
Dichloromethyl anion (C-HCl2)
C.
Dichlorocarbene (:CCl2)
Reimer-Tiemann reaction. The electrophile formed is :CCl2 (Dichlorocarbene) according to the following reaction
Carboxylic acids have higher boiling points than aldehydes, ketones and even alcohols of comparable molecular mass. It is due to their
Formation of intramolecular H-bonding
Formation of intermolecular H-bonding
Formation of intermolecular H-bonding
More extensive association of carboxylic acid via Vander Waals force of attraction
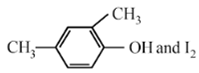
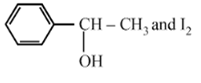
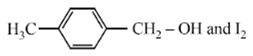
Compound A, C8H10O, is found to react with NaOI (produced by reacting Y with NaOH) and yields a yellow precipitate with characteristic smell. A and Y are respectively
![]()