Multiple Choice Questions
Multiple Choice QuestionsHydrocarbon (A) reacts with bromine by substitution to form an alkyl bromide which by Wurtz reaction is converted to a gaseous hydrocarbon containing less than four carbon atoms. (A) is
CH ≡ CH
H2C = CH2
CH4
CH3-CH3
In the reaction,
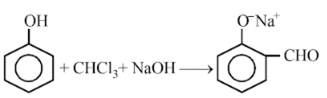
The electrophile involved is
Dichloromethyl cation (C+HCl2)
Formyl cation (C+HO)
Dichlorocarbene (:CCl2)
Dichloromethyl anion (C-HCl2)
Carboxylic acids have higher boiling points than aldehydes, ketones and even alcohols of comparable molecular mass. It is due to their
Formation of intramolecular H-bonding
Formation of intermolecular H-bonding
Formation of intermolecular H-bonding
More extensive association of carboxylic acid via Vander Waals force of attraction
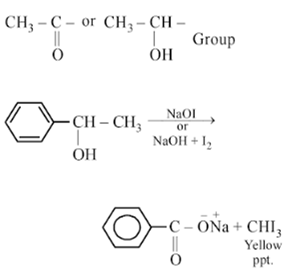
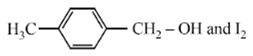
Compound A, C8H10O, is found to react with NaOI (produced by reacting Y with NaOH) and yields a yellow precipitate with characteristic smell. A and Y are respectively
![]()
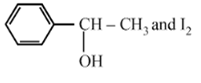
D.

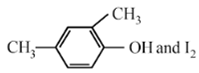
Haloform reaction is shown by compound having