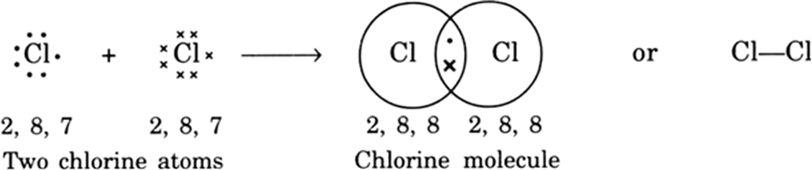
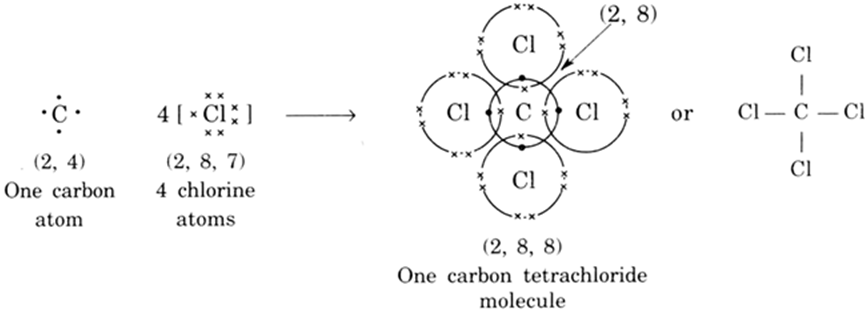
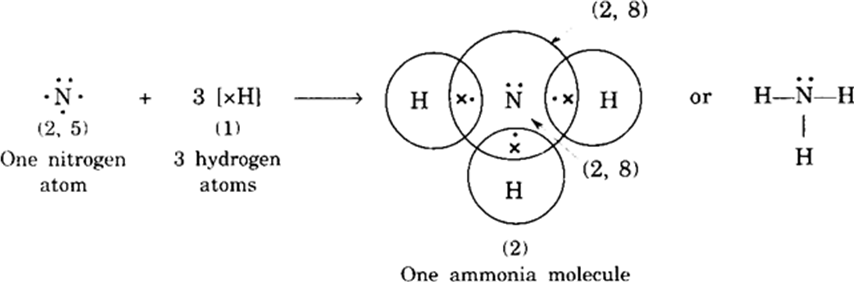
Discuss the reactivity of carbon in relation to its electronic configuration.
The electronic configuration of carbon is K (2), L (4). Thus it has 4 electrons in its outermost shell. In order to react or to achieve noble gas configuration, it should either gain or lose 4 electrons.
(i) It cannot gain 4 electrons and become C4– anion because its nucleus with 6 protons cannot hold on 10 electrons.
(ii) Also it cannot lose 4 electrons and become C4+ because a very large amount of energy would be required to remove four electrons from a small atom like carbon having a very close nucleus with 6 protons.
Carbon overcomes above problem and forms a large number of compounds by sharing its valence electrons with other atoms of carbon or with atoms of other elements, thereby satisfying requirement to attain noble configuration of all the combining atoms. For example, carbon forms methane as shown below: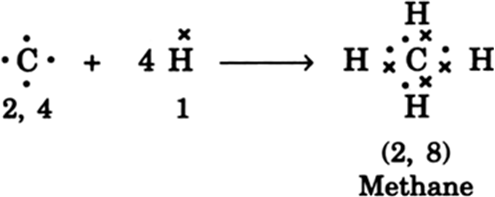
In methane, carbon shares its four valence electrons with four valence electrons, one each from four hydrogen atoms. Thus carbon attains the configuration 2, 8 as that of nearest noble gas, neon. And all four hydrogen atoms individually atttain the configuration of helium gas. Bond so formed due to sharing of electrons is called covalent bond.
