(2) Storage - The food is stored in the form of starch in plants and in the form of glycogen in animals.
(3)Structural support - Cellulose is the structural material of plants.
(4) Protective covering - Exoskeleton of the arthropods are made of polysaccharide chitin.
Amino acid are organic compounds made up of carbon, hydrogen, oxygen and nitrogen. They consist of an amino group and an acidic group attached to the the same carbon called α-carbon. Thus, they are also known as α-amino acids. They are substituted methanes. Four groups occupy the four valency positions. The four groups being an amino group, a hydrogen, A carboxylic group and a variable R group. The R group could be a hydrogen(the amino acid is called glycine), a methyl group (alanine) etc. There are 20 amino acids that occur in proteins.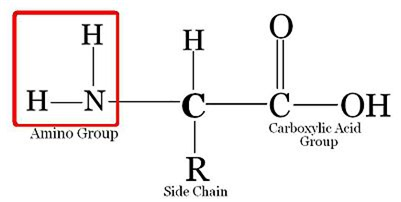
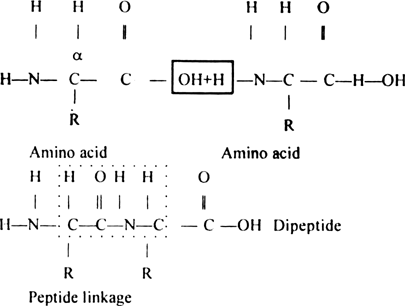
The resulting ![]() linkage is called a peptide linkage and the product is called dipeptide. The C–N bond in the peptide linkage is called a peptide bond.
linkage is called a peptide linkage and the product is called dipeptide. The C–N bond in the peptide linkage is called a peptide bond.
|
Unsaturated fatty acids |
Saturated fatty acids |
|
(1) They have one or two or three double bonds between the carbon atoms of the molecular chain. (2) They have lower melting and boiling points. (3) They cannot be synthesized in the body of any animal and are therefore essential. (4) Are more reactive and do not have a tendency to settle in the body. (5) Generally less harmful and do not cause cardiovascular diseases e.g. oleic acid.
|
(1) They have no double bonds between the atoms of the molecular chain. (2) They have higher melting points and boiling points. (3) They can be synthesized in the animal body and are therefore non-essential. (4) Are more stable and tend to accumulate in the body. (5) Harmfull and can cause cardiovascular diseases e.g. stearic acid.
|
1. Lipids act as important storage compounds.
2. They form membranous structures of the cell.
3. Lipids are rich source of energy and the energy produced by 1 gram of fat is more than twice than that produced from 1 gram of glucose.
4. They serve as main parts of cell wall in plants and form exoskeleton of insects.
5. In certain animals such as whale, polar bear etc. they form an insulating layer.
6. They are also used in the synthesis of steroid hormones and bile salts.
7. Fats from cushion like structures below vital organs such as brain, heart, kidneys, eyes etc. and protect them from mechanical shocks.
8. Fats insulate the nerve fibres electrically.
