
Mass of liquid = 100 gm
Density of liquid = 2 gm/cc
Volume of ballon containing liquid = 50cc
When it is fully immeresed in water, it will displace 50cc of water.
Therefore upward thrust on ballon is given by,
U = 50 x 1
= 50 gmf
Tension in string is,
T=100 - 50
= 50 gmf
(a) Ice floats in water with about nine-tenth of its volume submerged. What is the fractional volume submerged for an iceberg floating on a fresh water lake of (hypothetical) a planet whose gravity is ten times that of the earth?
(b) What is the fractional volume submerged of an ice-cube in a pail of water placed in an enclosure which is freely falling under gravity?
(a) Let a be the density of ice and ρ be the density of water.
The ice floats in water.
Therefore, weight of ice is equal to the weight of liquid displaced.
Let the volume of ice be V and volume of water displaced be V'.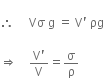
i.e. fractional volume displaced by ice is independent of g.
Therefore, on the new hypothetical planet, fractional volume displaced by ice is also same as that on the earth i.e. nine-tenth.
(b) In free falling state, ice-cube is in weightlessness state and also the thrust exerted by water is zero. Therefore, it can float with any volume submerged.
Fractional volume submerged,
