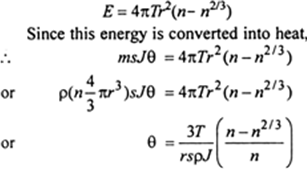
When we form the vertical film of soap solution, soap particles being heavy shift to lower side of film and hence surface tension on upper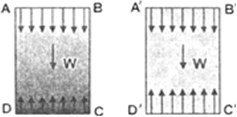
part is greater than the lower part which provides the necessary force to balance the weight of film, hence film is stable. But in vertical film of pure water surface tension forces are same at all the points i.e. on A' B' and C'D'. Therefore, a film of pure water will break in the absence of extra force required to balance the weight.
The surface area of liquid increases, when the sprayed in small droplets. Work has to be done against the cohesive force to increase the surface area. This work is done at the cost of internal energy and hence the temperature of drop falls.


