What will be the pressure inside a small air bubble of 0.1 mm radius situated just below the free surface of water?
(S.T. of water = 72dyne/cm; Atmospheric pressure 1.013 x 106 dyne/cm2 )
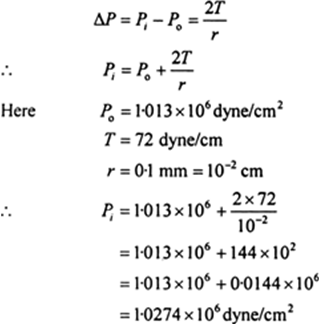
The different forces acting on the plate are:
(i) Weight W vertically downward,
(ii) Upward thrust U vertically upward,
(iii) Surface tension force T in downward direction .
These forces are given by,
Weight, W=8.6 x 980 = 8428 dyne
Upward Thrust, U=weight of water displaced
=Volume of water displaced x density of water x g
Let,
T = Total length of the water in touch with plate x surface tension
=2(1.5 + 0.2) x 70 = 238 dyne
Now the apparent weight,
Wa + W + T - U = 8428 + 238 - 1470
= 7196 dyne
= 7.434 gf
