
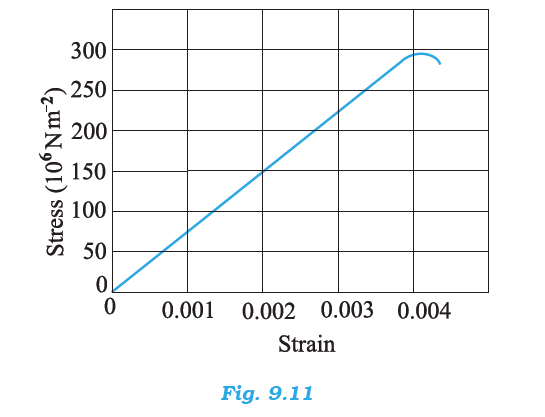
Read the following two statements below carefully and state, with reasons, if it is true or false.
(a) The Young’s modulus of rubber is greater than that of steel;
(b) The stretching of a coil is determined by its shear modulus.
a) False, because the modulus of elasticity is inversely proportional to the strain for a given stress. If steel and rubber are under the same stress, then strain in steel is less than rubber. Hence steel is more elastic than rubber.
(b) True, because the change takes place in the shape of the coil spring. Therefore, stretching is determined by its shear modulus.
Two wires of diameter 0.25 cm, one made of steel and the other made of brass are loaded as shown in Fig. 9.13. The unloaded length of steel wire is 1.5 m and that of brass wire is 1.0 m. Compute the elongations of the steel and the brass wires.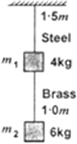
Given,
Diameter of the wires = 0.25 cm
That is, ds = dB = 0.25 cm
Therefore,
Radius of the wires will be given by,
Unloaded length of the steel wire, Ls=1.5 m
Unloaded length of the brass wire, LB = 1 m
Young's modulus of steel, Ys = 2 Pa
Young's modulus of brass, YB =
m1 = 4 kg and m2 = 6 kg
Brass wire is under a tension of load 6 kg.
Therefore, increase in length is given by
Since, brass wire is under a tension of load 6 kg.
"
Steel wire is under a tension of 10 kg load.
Therefore, elongation of length of the wire,

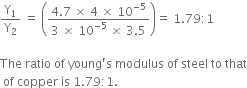
A steel wire of length 4.7 m and cross-sectional area 3.0 × 10-5 m2 stretches by the same amount as a copper wire of length 3.5 m and cross-sectional area of 4.0 × 10–5 m2 under a given load. What is the ratio of the Young’s modulus of steel to that of copper?
Length of steel wire, Ls = 4.7m
Length of copper wire, Lc=3.5m
Area of cross-section of steel wire, As=3.0 x 10-5 m2
Area of copper wire, Ac = 4.0 x 10-5 m2
To find = the ratio of Young's modulus of steel to that of copper?
