(a)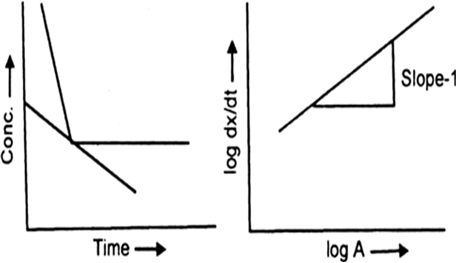
The rate i.e., dx / dt depends on the concentration of the reactant. Let dx / dt = k[A]1.
On taking log![]()
On plotting log dx / dt versus log A is always a straight line with slope equal to 1.
(b) Example of a first order reaction.
Decomposition of nitrogen pentoxide (N2O5)![]()
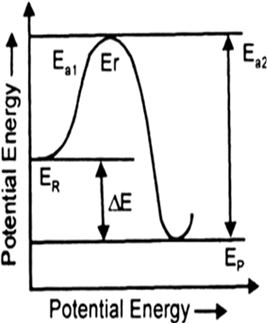
Reaction coordinate represents the profile of energy change when reactants change into products.
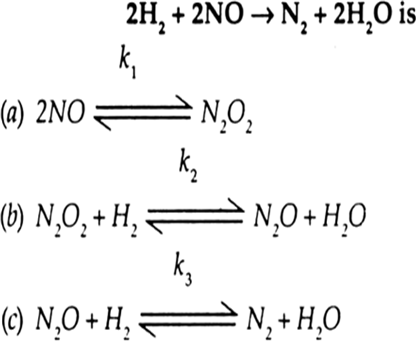
Energy of activation. The minimum energy over average energy which must be gained by the molecules before they could react to form products is called the energy of activation. It is denoted by Ea.
According to the Arrhenius theory, activation energy is independent of temperature. However, precise measurements indicate that the activation energy tends to decrease slightly with a rise in temperature.
Order of reaction with respect to Cl2 = 1.
Order of reaction with respect to NO = 2.
Overall order of reaction = 1 + 2 + 3.
