
|
exp.
|
[A]/ |
[B]/M |
Initial rate of formation |
|
I |
0.1 |
0.1 |
|
|
II |
0.3 |
0.2 |
7.2 x 10–2 |
|
III |
0.3 |
0.4 |
2.88 x 10–1 |
|
IV |
0.4 |
0.1 |
2.40 x 10–2 |
|
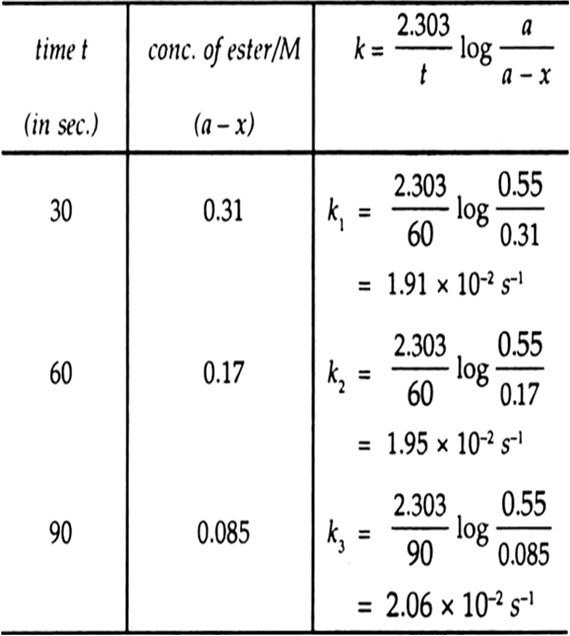
t / s |
0 |
30 |
60 |
90 |
|
Ester / mol L–1 |
0.55 |
0.31 |
0.17 |
|
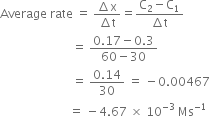
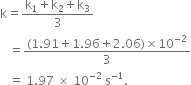
|
A/mol L–1 |
0.20 |
0.20 |
0.40 |
|
B/mol L–1 |
0.30 |
0.10 |
0.05 |
|
r/mol L–1S–1 |
5.07 x 10–5 |
5.07 x 10–5 |
1.43 x 10–4 |
What is the order of the reaction with respect of A and B?
In a reaction A and B, Let order of reaction w.r.t. A is x and w.r.t. B is y. Then the rate of reaction can be written as
rate = k[A]x [B]y
From given table data, 1 and 2 we can write
5.07 x 10–5 = k[10.20]x [0.30]y ...(i)
5.07 x 10–5 = k[0.20]x [0.10]y ...(ii)
Dividing (ii) by (i), we get
or
