
A secondary cell after use can be recharged by passing current through it in the opposite direction so that it can be used again. A good secondary cell can undergo a large number of discharging and charging cycles. The most important secondary cell is the lead storage battery commonly used in automobiles and invertors.
It consists of a lead anode and a grid of lead packed with lead dioxide (PbO2 ) as cathode. A 38% solution of sulphuric acid is used as an electrolyte.
The cell reactions when the battery is in use are given below:
Anode: Pb(s) + SO42–(aq)----> PbSO4(s) + 2e–
Cathode: PbO2(s) + SO42–(aq) + 4H+(aq) + 2e– ----> PbSO4 (s) + 2H2O (l )
i.e., overall cell reaction consisting of cathode and anode reactions is:
Pb(s) + PbO2(s) + 2H2SO4(aq)---> 2PbSO4(s) + 2H2O(l)
On charging the battery the reaction is reversed and PbSO4(s) on anode and cathode is converted into Pb and PbO2, respectively.
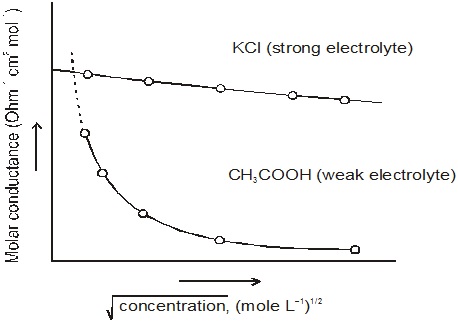
Kohlrausch’s law of independent migration of ions states molar conductivity of an electrolyte at infinite dilution can be expressed as the sum of the contribution of individual ions. If molar conductivity of cations and anions are represented by λ∞+ and λ∞– respectively.
where v+ and v– are number of cations and anions per formula of electrolyte e.g.,
Λ∞ CaCl2 = λ∞ (Ca2+) + 2 λ∞ (CI–)
Λ = KCl = λ∞ (K+) + λ∞ (CI–)
Uses 1. It is used to find molar conductivity of weak electrolyte at infinite dilution which
cannot be obtained by extrapolation.
2. It is used to calculate degree of dissociation of weak electrolyte at a particular concentration.
Degree of dissociation
where Λm is molar conductivity of weak electrolyte at a particular concentration and Λemis molar conductivity of weak electrolyte at infinite dilution.
Gibbs energy of the reaction given by:
= – nFE(cell)
thus the reaction
Zn(s) + Cu2+(aq)---> Zn2+(aq) + Cu(s)
= – 2FE(cell)
but when we write the reaction
2 Zn (s) + 2 Cu2+----->2 Zn2+(aq) + 2Cu(s)
= – 4FE(cell)
If the concentration of all the reacting species is unity, then
E(cell) =
and we have
= – nF
Thus, from the measurement of we can obtain an important thermodynamic quantity, , standard Gibbs energy of the reaction.
From the latter we can calculate equilibrium constant by the equation:
= –RT ln Kc
