
From the table, standard electrode potents at 298 k are:
(E°Fe3+/VF = 0.77 V, E°I2/I– = 0.54 V)
(E°Ag+/Ag = E°Cu2+/Cu = 0.34)
(E°Fe3+/Fe= 0.77 V, E°Br2/Br- = 1.08 V)
(E°Ag+/Ag= 0.8 V E°Fe3+/Fe2+ = 0.77 V)
(E°Fe3+/Fe2+= 0.77 V,E°Br2/Br- = 1.08 V)
(a) ![]()
In this reaction, Fe3+ is reduced to Fe2+ and I– is oxidised to I2. The cell giving above reaction will be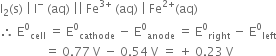
As E0 is positive, the reaction between Fe3+ (aq) and I– (aq) occurs as indicated by possible reaction given above.
(b) ![]()
Here, in this reaction, Ag+ is reduced to Ag (i.e., it should be cathode) and Cu(s) is oxidised to Cu2+(aq) (i.e., it should be anode).
The cell can be represented as

As E°cell is positive, the reaction between (Ag+ (aq) and Cu(s) occurs as indicated by possible reaction given above.
(c) ![]()
In this reaction Fe3+ is reduced to Fe2+ (i.e., Fe3/Fe2+ electrode should be cathode) and Br is oxidised to Br2 (i.e., Br2/Br– electrode should be anode.
The cell can be represented as: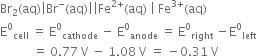
As E°cell is negative, no reaction will occur between Fe3+ (aq) and Br–(aq).
(d) ![]()
Two half-cell reactions can be expressed as: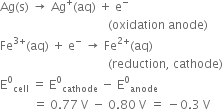
As E°cell is negative, no reaction occurs between Fe3+(aq) and Ag(s).
(e) ![]()
The two half-cell reactions are
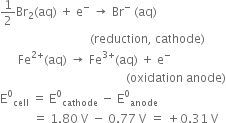
As E°cell is positive, the reaction is feasible, i.e., reaction between Br2(aq) and Fe2+ (aq) occurs as indicated by possible reaction given above.
