For cell![]()
The cell reaction
![]()
![]()
On applying Nernst equation
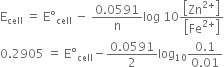
or ![]()
or ![]()
or ![]()
At equilibrium ![]()
![]()
∴ ![]()
or ![]()
or ![]()
or ![]()
The following electrochemical cell has been set up
Pt(1) | Fe3+, Fe2+ (a = 1) || Ce4+, Ce3+ (a = 1) | Pt (2)
E°(Fe2+) = 0.77 V, and E°(Ce4+,Ce3+) = 1.61 V
If an ammeter is connected between the two platinum electrodes, predict the direction of flow of current. Will the current increase or decrease with time?
For the electrochemical cell
Pt(1) | Fe3+, Fe2+ (a = 1) || Ce4+, Ce3+ (a = 1) | Pt (2)
the cell regions are
Right-half cell: reduction
Left-half cell: oxidation
______________________________
Add
The net cell potential is
E° Cell = E° R – E° L = 1.61 V – 0.77 V = 0.84 V.
Since E°cell is positive, the cell reaction will be spontaneous.
The current in the external circuit will flow from Pt (1) (which serves as anode to Pt(2) which serves as cathode.
With the passage of time, Ecell will decrease and so is the current in the external circuit.
Resistance of KCl solution,
R = 85
Cell constant = K x R
Resistance of unknown electrolyte solution,
Specific conductance
Concentration,
Molar conductance,
