Calculate the cell emf at 25° C for the following cell:
Ni(s) | Ni2+ (0.01 M) || Cu2+ (0.1 M) | Cu(s)
[Given E°Ni2+/Ni = – 0 25 V, E° Cu = + 0.34 V, 1 F = 96500]
Calculate the maximum work that can be accomplished by operation of this cell.
Ni(s) | Ni2+ (0.01 M) || Cu2+ (0.1 M) | Cu(s)
At anode Ni(s) → Ni2+ (aq) + 2e
At cathode Cu2+ + 2e → Cu(s)
Net cell reaction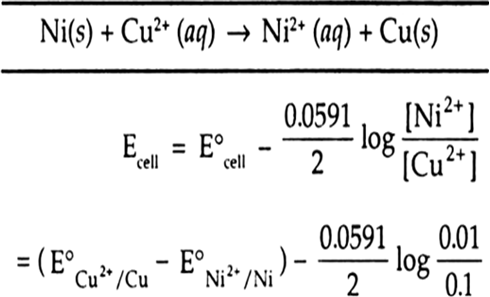
The emf of a cell corresponding to the reaction
Zn(s) + 2H+ (aq) → Zn2+ (0.1 M) + H2(g) (1 atm) is 0.28 V at 15° C.Write the half cell reactions and calculate the pH of the solution at the hydrogen electrode.
Mg(s) | Mg2+ (0.01 M) || Sn2+ (0.1 M) | Sn(s)
Mg(s) → Mg2+ (aq) + 2e–
(oxidation at anode)
Sn2+(aq) + 2e– → Sn(s)
(reduction at cathode)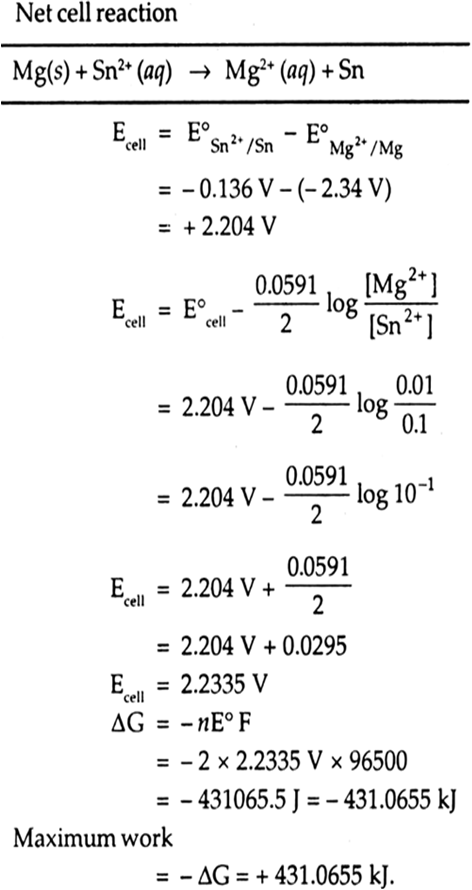
(a)
(b) Now,
