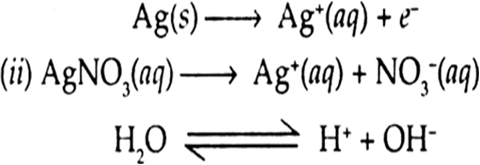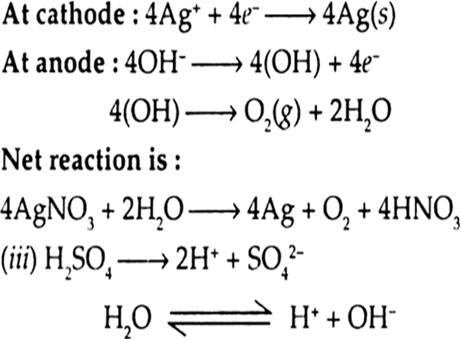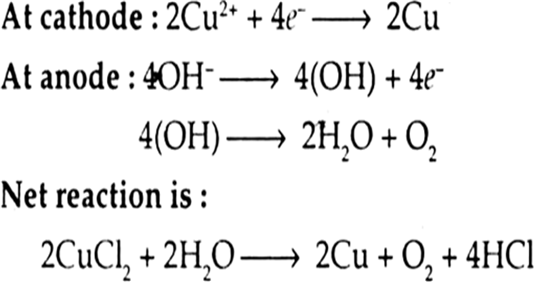Depict the galvanic cell in which the reaction
Zn(s) + 2Ag+(aq) → Zn2+(aq) + 2Ag(s) takes place. Further show:
(i) Which of the electrode is negatively charged?
(ii) The carriers of the current in the cell.
(iii) Individual reaction at each electrode.

(ii) Electrons move from anode (zinc electrode) to cathode (silver electrode) in the external circuit. Zinc ions go into solution at anode and Ag+ ions get deposited at cathode. Thus electrons in the external and metal ions in the internal circuit act as carrier of current in.
(iii) Overall reaction is obtained by anode and cathode reactions.
Zn(s) + 2Ag+(aq) → Zn2+(aq) + 2Ag(s)
Predict the products of electrolysis in each of the following:
(i) An aqueous solution of AgNO3 with silver electrodes.
(ii) An aqueous solution of AgNO3 with platinum electrodes.
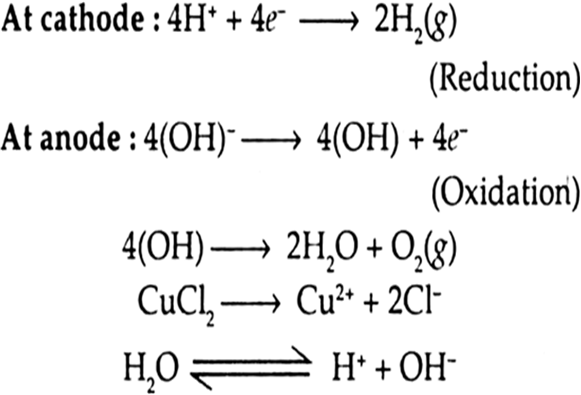
(iii) A dilute solution of H2SO4 with platinum electrodes.
(iv) An aqueous solution of CuCl2 with platinum electrodes.