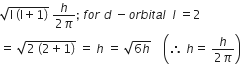Magnetic moment 2.84 BM is given by (At. no. Ni= 28, Ti= 22, Cr=24, Co = 27)
Ni2+
Ti3+
Cr3+
Cr3+
A.
Ni2+
What is the maximum number of orbitals that can be identified with the following quantum numbers?
n=3, l =1, m1 = 0
1
2
3
3
A.
1
The value of n=3 and l =1 suggest that it is a 3p orbital while the value of m1 = 0 [magnetic quantum number] shows that the given 3p orbital is 3pz in nature.
Hence, the maximum number of orbitals identified by the given quantum number is only 1, i.e. 3pz.
Two electrons occupying the same orbital are distinguished by
Magnetic quantum number
Azimuthal quantum number
Spin quantum number
Spin quantum number
C.
Spin quantum number
Two electrons occupying the same orbital has equal spin but the directions of their spin are opposite. Hence, spin quantum number, s(represented +1/2 and -1/2) distinguishes them.
The angular momentum of electrons in d orbital is equal to




A.

Angular momentum of electrons in d- orbital is