 Multiple Choice Questions
Multiple Choice QuestionsRed precipitate is obtained when ethanol solution of dimethylglyoxime is added to ammoniacal Ni (II).Which of the following statements is not true?
Red Complex has a square planar geometry
Complex has symmetrical H- bonding
Red complex has a tetrahedral geometry
Red complex has a tetrahedral geometry
C.
Red complex has a tetrahedral geometry
The reaction of Nickel with DMG Gives,
It shows that DMG acts as a bidentate ligand.
also, the geometry of DMG is square planar.
Hybridization of DMGi is dsp2 thus structure of is square planar.
Low spin complex of d6 -cation in an octahedral field will have the following energy
(Δo = Crystal field splitting energy in an octahedral field,
P= electron pairing energy)
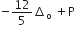

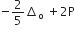
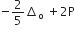
Chloramphenicol is an
antifertility drug
antihistamine
antiseptic and disinfectant
antiseptic and disinfectant
Which of the following compounds can be used as antifreeze in automobile radiators?
Methyl alcohol
Glycol
Nitrophenol
Nitrophenol
Which of the following reagents will be able to distinguish between 1-butyne and 2-butyne?
NaNH2
HCl
O2
O2
Consider the reaction,
RCHO +NH2NH2 →RCH =N-NH2
What sort of reaction is it?
Electrophilic addition elimination reaction
Free radical addition-elimination reaction
Electrophilic substitution elimination reaction
Electrophilic substitution elimination reaction
Which one of the following sets forms the biodegradable polymer?
CH2 =CH-CN and CH2 =CH-CH=CH2
H2N-CH2-COOH and H2N-(CH2)5-COOH
HO-CH2-CH2-OH and ![]()
HO-CH2-CH2-OH and ![]()
An organic compound (C3H9N) (A), when treated with nitrous acid, gave an alcohol and N2 gas was evolved. (A) on warming with CHCl3 and Caustic potash gave (C) which on reduction gave iso-propyl methylamine predict the structure of (A)
CH3-CH2 -NH -CH3
Which of the following compounds will give a yellow precipitate with iodine and alkali?
Acetophenone
Methyl acetate
Acetamide
Acetamide
