Give simple chemical tests to distinguish between the following pairs of compounds:
(i) Ethanal and Propanal
(ii) Benzoic acid and Phenol
(i) Distinguish test between ethanal and propanal:
Iodoform Test: Ethanal gives iodoform test.
CH3CHO + 4NaOH + 3I2 ---> CHI3 (Yellow ppt.) + HCOONa + 3NaI + 3H2O
Propanal does not give this test.
CH3CH2CHO + 4NaOH + 3I2 ---> No Reaction.
(ii) Distinguish test between Benzoic acid and Phenol:
NaHCO3Test: When Benzoic acid reacts with NaHCO3, the brisk effervescence of CO2 gas evolved.
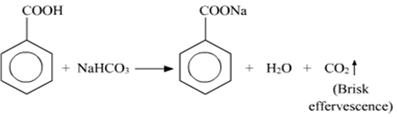
Phenol does not give this test.
C6H5OH + NaHCO3 --> No Reaction
A solution of (–) –1 – chloro –1 – phenylethane is toluene racemises slowly in the presence of a small amount of SbCl5, due to the formation of
-
carbanion
-
Carbene
-
Carbocation
-
Carbocation
Arrange the following compounds in order of decreasing acidity
-
II>IV>I>III
-
I>II>III>IV
-
III>I>II>IV
-
III>I>II>IV
An aromatic compound 'A' of molecular formula C7H7ON undergoes a series of reactions as shown below. Write the structures of A, B, C, D and E in the following reactions: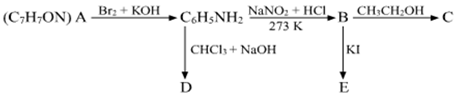
Select the ether among following that yields methanol as one the products on reaction with cold hydroiodic acid
1-methoxybutane
1-methoxybutane-2-methylpropane
2-methoxy-2-methylpropane
methoxybenzene