CBSE
Class 10 Class 12
Heterogeneous mixtures can be separated into their respective constituents by simple physical methods like handpicking, sieving, filtration.
Sometimes special techniques have to be used for the separation of the components of a mixture.
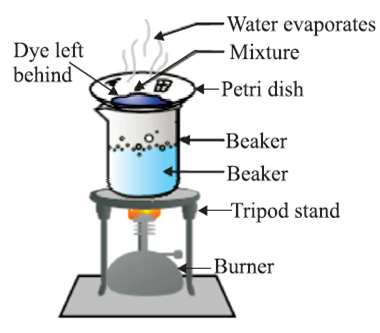
By using Evaporation method, we can obtain coloured component from blue/black ink.
Basic principle: Out of the two components of a mixture one can evaporate [i.e., has less boiling point] and other has a higher boiling point.
Example: Mixture of dye [higher boiling point] and water. Out of water and dye, water evaporates but the dye is left behind in a petri dish.
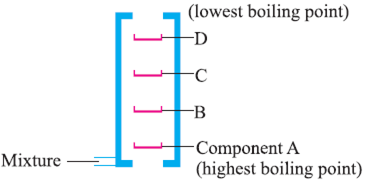
Fractional Distillation of Air:
Air is also separated by this method.

Some of the applications of fractional distillation :
Crystallisation Method :
Basic principle: To remove impurities from a mixture by first dissolving in a suitable solvent and then crystallising out one component.
For example, Copper sulphate crystals (impure) are first dissolved in sulphuric acid and then heated to a saturated solution. Now, this solution is left overnight. So, only pure copper sulphate crystals are formed whereas impurities are left behind in the solution. This solution can be thus filtered so as to get pure copper sulphate crystals on filter paper.
Crystallisation better than evaporation:
Applications :
Sublimation Method:
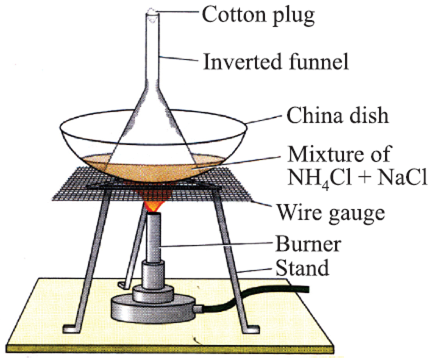
Basic principle: Out of the two components, one will sublime (directly converts to gas from solid) and others will not.
Example: NH4Cl (ammonium chloride) and NaCl common salt mixture can be easily separated by heating so that NH4Cl sublimes but common salt remains behind.
Applications :
Camphor, naphthalene, anthracene, NH4Cl can sublime.
Differential extraction method :
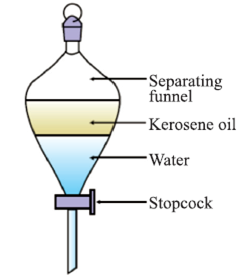
Basic principle: Two immiscible liquids (which do not dissolve in each other) can be easily separated by putting in a differential extraction funnel/ separating funnel.
Example: Water from oil can be separated by first opening the stopcock till water is removed in one beaker, then afterwards oil can be collected in a separate beaker.
Applications :
Distillation Method:
Principle: Out of the two components one has a lower boiling point and other has a higher boiling point. This is used to separate two or more miscible liquids.
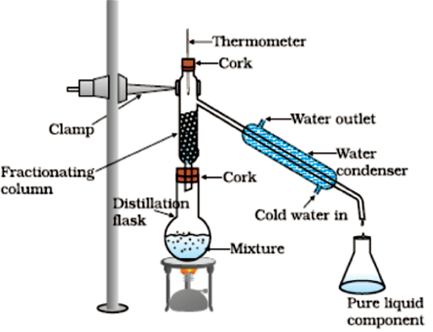
Example: When a mixture of acetone and water is heated, acetone having a lesser boiling point, boils and moves to the delivery tube, within which it condenses back to liquid with the help of a condenser clamped to it. Thus, acetone is separated out in a beaker and water is left in the distillation flask.
If there are more than two components (liquids) mixed (with different boiling points) then we use a fractionating column to separate all the components from each other. This process is done for air, petroleum etc.
Petroleum is separated into paraffin wax, lubricating oil, diesel, kerosene, petrol and petrol gas by this method.
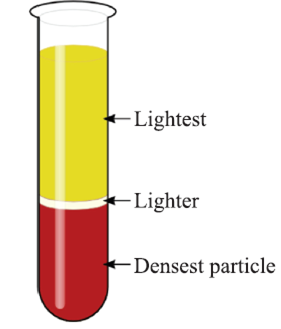
By using Centrifugation method, we can separate cream from milk.
Basic principle: When the mixture is rotated very fast, then denser particles are forced at the bottom and lighter particles stay above.
For example, Separating cream from milk.
Applications :
Chromatography Method:
Basic principle: Coloured components of a mixture can be separated by using an Absorbent on which they are absorbed at different rates.
(Absorption is the process of surface absorption)
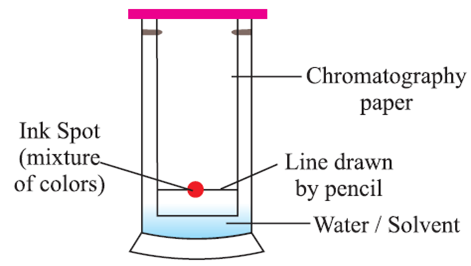
When water/any suitable solvent moves up, the chromatography paper ink with two different colours separates because both colours are absorbed at different speeds.
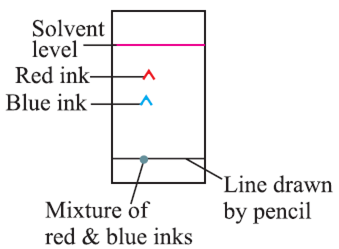
Applications :