 Multiple Choice Questions
Multiple Choice QuestionsMatch List – I (Fundamental Experiment) with List – II (its conclusion) and select the correct option from the choices given below the list:
List I |
List II |
| (A) Franck-Hertz Experiment | (i) Particle nature of light |
| (B) Photo-electric experiment | (ii) Discrete energy levels of atom |
| (C) Davison – Germer Experiment | (iii) Wave nature of electron |
| (iv) Structure of atom |
| A | B | C |
| 1 | 4 | 3 |
| A | B | C |
| 2 | 4 | 3 |
| A | B | C |
| 2 | 1 | 3 |
| A | B | C |
| 2 | 1 | 3 |
The anode voltage of a photocell is kept fixed. The wavelength λ of the light falling on the cathode is gradually changed. The plate current I of the photocell varies as follows:
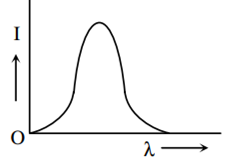
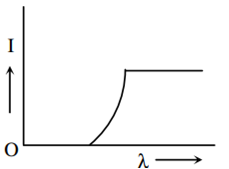
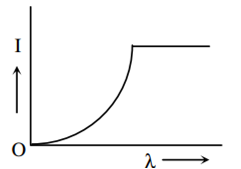

This question has Statement 1 and statement 2. Of the four choices given after the statements, choose the one that best describes the two statements :
Statement 1 :
A metallic surface is irradiated by a monochromatic light of frequency v > v0 (the threshold frequency). The maximum kinetic energy and the stopping potentials are Kmax and V0 respectively. If the frequency incident on the surface is doubled, both the Kmax and V0 are also doubled.
Statement 2 :
The maximum kinetic energy and the stopping potential of photoelectrons emitted from a surface are linearly dependent on the frequency of incident light.
Statement 1 is true, statement2 is false.
Statement 1 is true, Statement 2 is true, Statement 2 is the correct explanation of Statement 1
Statement 1 is true, Statement 2 is true, Statement 2 is not the correct explanation of Statement 1
Statement 1 is true, Statement 2 is true, Statement 2 is not the correct explanation of Statement 1
STATEMENT – 1
When ultraviolet light is incident on a photocell, its stopping potential is V0 and the maximum kinetic energy of the photoelectrons is Kmax. When the ultraviolet light is replaced by X–rays, both V0 and Kmax increase.
STATEMENT – 2
Photoelectrons are emitted with speeds ranging from zero to a maximum value because of the range of frequencies present in the incident light.
Statement – 1 is True, Statement – 2 is False.
Statement – 1 is True, Statement – 2 is True; Statement – 2 is a correct explanation for Statement – 1.
Statement – 1 is True, Statement – 2 is True; Statement – 2 is not the correct explanation for Statement – 1.
Statement – 1 is True, Statement – 2 is True; Statement – 2 is not the correct explanation for Statement – 1.
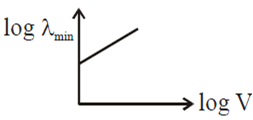
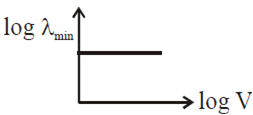
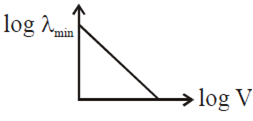
An electron beam is accelerated by a potential difference V to hit a metallic target to produce X-rays. It produces continuous as well as characteristic X-rays.If λmin is the smallest possible wavelength of X-ray in the spectrum, the variation of log λ min with log V is correctly represented in

In a Young's double slit experiment, slits are separated by 0.5 mm, and the screen is placed 150 cm away. A beam of light consisting of two wavelengths, 650 nm and 520 nm, is used to obtain interference fringes on the screen. The least distance from the common central maximum to the point where the bright fringes due to both the wavelengths coincide is
9.75 mm
15.6 mm
1.56 mm
1.56 mm
The surface of a metal is illuminated with the light of 400 nm. The kinetic energy of the ejected photoelectrons was found to be 1.68 eV. The work function of the metal is (hc = 1240 eV nm)
3.09 eV
1.41 eV
151 eV
151 eV
Wave property of electrons implies that they will show diffraction effects. Davisson and Germer demonstrated this by diffracting electrons from crystals. The law governing the diffraction from a crystal is obtained by requiring that electron waves reflected from the planes of atoms in a crystal interfere constructively (see in the figure).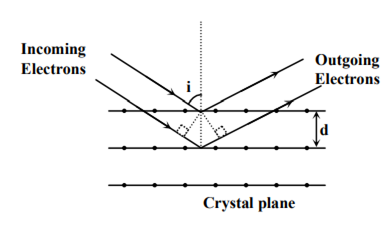
If a strong diffraction peak is observed when electrons are incident at an angle ‘i’ from the normal to the crystal planes with distance’ between them (see figure), de Broglie wavelength λdB of electrons can be calculated by the relationship (n is an integer)
d sin i = nλdB
2d cos i = nλdB
2d sin i = nλdB
2d sin i = nλdB
Wave property of electrons implies that they will show diffraction effects. Davisson and Germer demonstrated this by diffracting electrons from crystals. The law governing the diffraction from a crystal is obtained by requiring that electron waves reflected from the planes of atoms in a crystal interfere constructively (see in the figure).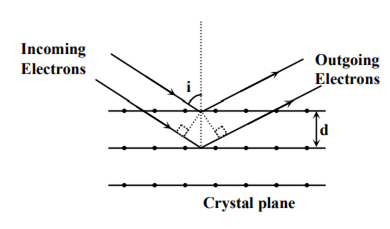
In an experiment, electrons are made to pass through a narrow slit of width’ comparable to their de Broglie wavelength. They are detected on a screen at a distance ‘D’ from the slit (see figure). 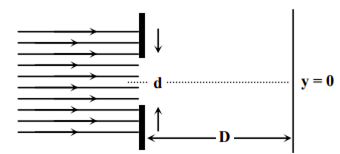
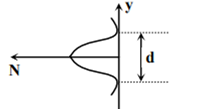
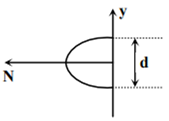
Which of the following graph can be expected to represent the number of electrons ‘N’ detected as a function of the detector position ‘y’(y = 0 corresponds to the middle of the slit)?
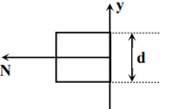

A photon of frequency v has a momentum associated with it. If c is the velocity of light, the momentum is
v /c
h v c
h v /c2
h v /c2
