 Multiple Choice Questions
Multiple Choice QuestionsUsing the Gibbs energy change, ΔG0 = +63.3 KJ for the following reaction,
Ag2CO3 (s) r2Ag+ (aq) + CO32- (aq)
the Ksp of Ag2CO3 (s) in water at 250 C is (R= 8.314 JK-1 mol-1)
3.2 x 10-26
8.0 x 10-12
2.9 x 10-3
2.9 x 10-3
B.
8.0 x 10-12
At 250 molar conductance of 0.1 molar aqueous solutions of ammonium hydroxide is 9.54 ohm-1 cm2 mol-1 and at infinite dilution, its molar conductance is 238 ohm-1 cm2 mol-1. The degree of ionisation of ammonium hydroxide at the same concentration and temperature is
2.080%
20.800%
4.008%
4.008%
A button cell used in watches functions as following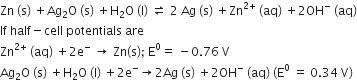
The cell potential will be
1.10 V
0.42 V
0.84 V
0.84 V
Standard reduction potentials of the half-reactions are given below.
F2 (g) +2e- → 2F- (aq) ; Eo = +2.85 V
Cl2 (g) +2e- →2Cl- (aq) ; Eo = +1.36V
Br2 (l) +2e- → 2Br- (aq) ; Eo = +1.06 V
I2 (s) +2e- →2I- (aq); Eo = +0.53 V
The strongest oxidising and reducing agents respectively are
F2 and I-
Br2 and Cl-
Cl2 and Br-
Cl2 and Br-
Molar conductivities (Λom) at infinite dilution of NaCl, HCl and CH3COONa are 126.4, 425.9 and 91.0 S Cm2 mol-1 respectively. Λom for CH3COOH will be
425.5 S cm2 mol-1
180.5 S cm2 mol-1
290.8 S cm2 mol-1
290.8 S cm2 mol-1
The Gibb's energy for the decomposition of Al2O3 at 500o C is as follow
2/3 Al2O3 → 4/3 Al + O2;
ΔrG = +960 kJ mol-1
The potential difference needed for the electrolytic reduction aluminium oxide (Al2O3) at 5000 C is at least
4.5 V
3.0 V
2.5 V
2.5 V
Standard electrode potential of three metal X, Y and Z are -1.2 V, +0.5 V and -3.0 V respectively. The reducing power of these metals will be
Y > X > Z
Z> X> Y
X > Y > Z
X > Y > Z
If the Eocell for a given reaction has a negative value then which of the following gives the correct relationships for the values of ΔGo and Keq ?
ΔGo < 0; Keq > 1
ΔGo < 0; Keq < 1
ΔGo > 0; Keq < 1
ΔGo > 0; Keq < 1
The electrode potentials for
Cu2+ (aq) + e- → Cu+ (aq) and Cu+ (aq) + e- →Cu (s)
are +0.15 V and +0.50V respectively. The value of  will be
will be
0.325 V
0.650 V
0.150 V
0.150 V
