 Multiple Choice Questions
Multiple Choice QuestionsThe step which never be involved in the dehydration of alcohol is
protonation
elimination
hydride transfer
carbanion formation
The boiling point of ethanol is higher as compared to the boiling point of diethyl ether though both have the same molecular formula. This is due to
resonance
-R group
H-bonding
covalent bonding
Which one of the following compounds will not react with CH3MgBr ?
Ethyl acetate
Acetone
Dimethyl ether
Ethanol
Phenol reacts with dilute nitric acid at normal temperature to form
o-nitrophenol
m-nitrophenol
o-and p-nitropheno
2, 4, 6-trinitrophenol
Which of the following compounds does not give Friedel-Craft's reaction ?
benzene
xylene
nitrobenzene
phenol
C.
nitrobenzene
Friedel - Craft's reaction involves electrophilic substitution and electrophile attacks at the electron rich site. Presence of electron releasing substituent (like CH3) increases the electron density over benzene nucleus, and thus activates it towards electrophlc substitution. -NO2 is a electron withdrawing group and thus, deactivates the benzene nucleus strongly. Hence, nitrobenzene does not give Friedel-Craft's reaction.
Which one is the correct IUPAC name of the following compound?

1-chloro-5-hydroxycydohexane
2-chloro-4-hydroxycyclohexane
3-chloro-3-cyclohexenol
5-hydroxycydohexenyl chloride
Which of the following represents the correct decreasing order of acidity of the following compounds?
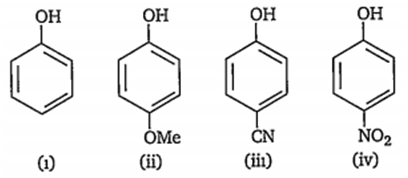
ii > i > iii > iv
iv > iii > ii > i
iii > iv > i > ii
iv > iii > i > ii
How much ethyl alcohol C2H5OH, must be added to 1.00 L of water so that the solution will not freeze at - 4°F?
211 gm
495 gm
85 gm
46 gm
What will be the major product when 2-amino propane is treated with nitrous acid?
Propane-2-ol
Cyclopropene
Propanol
2-rntropropane
