 Multiple Choice Questions
Multiple Choice QuestionsThe two structures written below represent
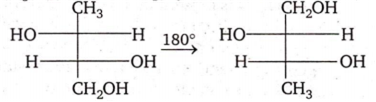
pair of diastereomers
pair of enantiomers
same molecule
both are optically inactive
A compound is formed by substitution of two chlorine for two hydrogens in propane. The number of possible isomeric compounds is
4
3
2
5
1 mole of methyl amine on reaction with nitrous acid gives at NTP
1.0 L of nitrogen
22.4 L of nitrogen
11.2 L of nitrogen
5.6 L of nitrogen
The major product P in the following reaction is
CH3-CH=CH2 P
CH3CH2CH2I
CH3-CH(I)-CH3
CH2(I)-CH=CH2
CH2(I)-CH2(I)
Which of the following will exhibit cis-trans isomerism?
CH2Br-CH2Br
CBr3 - CH3
CHBr = CHBr
CBr2= CH2
The IUPAC name of CH3-CH=CH-C≡CH is
pent-3-en-1-yne
pent-3-en-4-yne
pent-2-en-4-yne
pent-2-en-3-yne
The correct IUPAC name of ![]() is
is
1-cyclopropyl cyclobutane
1, 1-dicyclobutane
1-cyclobutane- 1- cyclopropane
none of the above
Geometrical isomerism is possible in
acetone- oxime
isobutene
acetophenone-oxime
benzophenone-oxime
IUPAC name of the following compound is:
CH3CH2C(Br) = CH - Cl
2-bromo-1-chloro butene
1-chloro-2-bromo butene
3-chloro-2-bromo butene-2
none of the above
