 Multiple Choice Questions
Multiple Choice Questions1,4-dimethylbenzene on heating with anhydrous AlCl3 and HCl produces
1, 2-dimethylbenzene
1, 3-dimethylbenzene
1,2, 3-trimethylbenzene
ethylbenzene
Best reagent for nuclear iodination of aromatic compounds is
KI/CH3COCH3
I2/CH3CN
KI/ CH3COOH
I2/ HNO3
The IUPAC name of the following molecule is
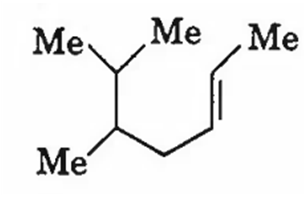
5,6-dimethylhept-2-ene
2,3-dimethylhept-5-ene
5,6-dimethylhept-3-ene
5-iso-propylhex-2-ene
The best method for the preparation of 2, 2-dimethylbutane is via the reaction of
Me3CBr and MeCH2Br in Na/ether
(Me3C)2CuLi and MeCH2Br
(MeCH2)2CuLi and Me3CBr
Me3CMgI and MeCH2I
The ease of nitration of the following three hydrocarbons follows the order
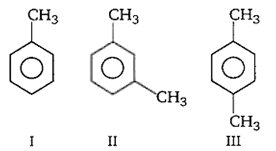
II - III ≈ I
II > III > I
III > II > I
I = III > II
Friedel-Craft's reaction using MeCl and anhydrous AlCl3 will take place most efficiently with
benzene
nitrobenzene
acetophenone
toluene
D.
toluene
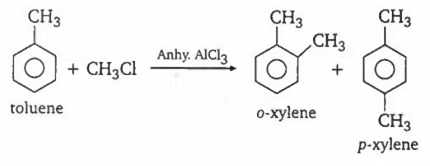
Friedel-crafts reaction is an electrophilic substitution reaction and presence of an electron releasing substituent like Me, makes the benzene nucleus more reactive towards such reactions. Thus toluene is most reactive among the given.
