 Multiple Choice Questions
Multiple Choice QuestionsThe compound that would produce a nauseating smell/ odour with a hot mixture of chloroform and ethanolic potassium hydroxide is
PhCONH2
PhNHCH3
PhNH2
PhOH
C.
PhNH2
When primary amine reacts with chloroform and ethanolic potassium hydroxide, they produce isocyamides as main product. The reaction is also known as carbylamine reaction.
Ph-NH2 Ph.N → C
The correct statement regarding the following compounds is
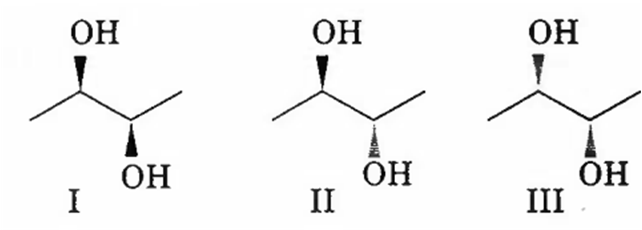
all three compounds are chiral
only I and II are chiral
I and III are diastereomers
only I and III are chiral
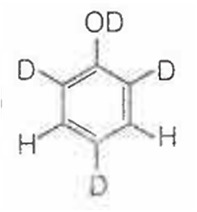
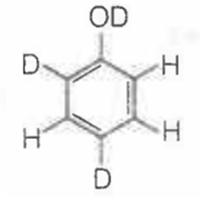
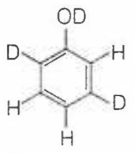
When phenol is treated with D2SO4 /D2O, some of the hydrogens get exchanged.The final product in this exchange reaction is
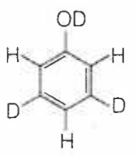
The correct order of acid strength of the following substituted phenols in water at 28°C is
p-nitrophenol < p-fluorophenol < p-chlorophenol
p-chlorophenol < p-fluorophenol < p-nitrophenol
p-fluorophenol < p-chlorophenol < p-nitrophenol
p-flurophenol < p-nitrophenol < p-chlorophenol
Correct statement(s) in cases of n-butanol and t-butanol is (are)
both are having equal solubility in water
t-butanol is more soluble in water than n-butanol
boiling point of t-butanol is lower than n-butanol
boiling point of n-butanol is lower than t-butanol
