 Multiple Choice Questions
Multiple Choice QuestionsIn the disproportionation reaction,
3HClO3 → HClO4 + Cl2 + 2O2 + H2O
the equivalent mass of the oxidising agent is (molar mass of HClO3 = 84.45)
16.89
32.22
84.45
28.15
A.
16.89
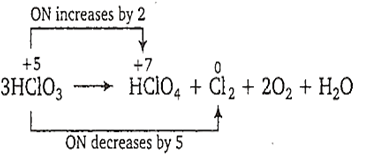
This is a disproportionation reaction since here the oxidation state of chlorine decreases from +5 to 0 in Cl2 as well as increases from +5 to +7 in HClO3. Thus, HClO3 acts as oxidising as well as reducing agent.
Equivalent mass of oxidising agent (i.e., HClO3)
=
= = 16.89
To an aqueous solution containing anions a few drops of acidified KMnO4 are added. Which one of the following anions, if present will not decolourise the KMnO4 solution ?
I-
CO
S2-
NO
In the following reaction,
Mx+ + MnO → MO + Mn2+ + O2
If one mole of MnO oxidises 2.5 moles of Mx+, then the value of x is
5
3
2
1
When MnO2 is fused with KOH, a coloured compound is formed. The product and its colour is
K2MnO4, purple green
KMnO4, purple
Mn2O3, brown
Mn2O4, black
