 Multiple Choice Questions
Multiple Choice QuestionsThe statement that is INCORRECT about the interstitial compounds is :
they are chemically reactive
they have metallic conductivity
they have high melting points
they are very hard
Consider the bcc unit cells of the solids 1 and 2 with the position of atoms as shown below. The radius of atom B is twice that of atom A. The unit cell edge length is 50% more in solid 2 than in 1. What is the approximate packing efficiency in solid 2?
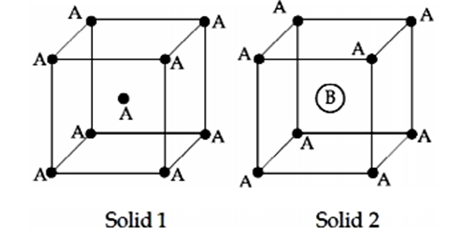
75%
65%
45%
90%
