 Multiple Choice Questions
Multiple Choice QuestionsThe electronic configuration of Eu (Atomic no.63), Gd (Atomic no. 64) and Tb (Atomic no. 65) are
[Xe]4f6 5d1 6s2, [Xe] 4f7 5d1 6s2 and [Xe] 4f9 6s2
[Xe] 4f6 5d1 6s2 , [Xe] 4f7 5d1 6s2 and [Xe] 4f8 5d1 6s2
[Xe] 4f7 6s2, [Xe] 4f7 5d1 6s2 and [Xe] 4f9 6s2
[Xe] 4f7 6s2, [Xe] 4f7 5d1 6s2 and [Xe] 4f9 6s2
In which of the following options the order of arrangement does not agree with the variation of the property indicated against it?
B<C<N<O (increasing first ionisation enthalpy)
I<Br<Cl<F(increasing electron gain enthalpy)
Li<Na<K<Rb (increasing metallic radius)
Li<Na<K<Rb (increasing metallic radius)
The species Ar, K+ and Ca2+ contain the same number of electrons. In which order do their radii increase?
Ar < K+< Ca2+
Ca2+ < Ar < K+
Ca2+ < K+ < Ar
Ca2+ < K+ < Ar
Solubility of the alkaline earth's metal sulphates in water decreases in the sequence
Mg>Ca>Sr>Ba
Ca>Sr>Ba>Mg>
Sr>Ca>Mg>Ba
Sr>Ca>Mg>Ba
Which of the following pairs of ions are isoelectronic and isostructural?
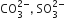
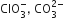


D.

Isostructural chemical compounds have similar chemical structures.
Isoelectronic terms refer to two atom, ions or molecules that have the same number of valence electrons.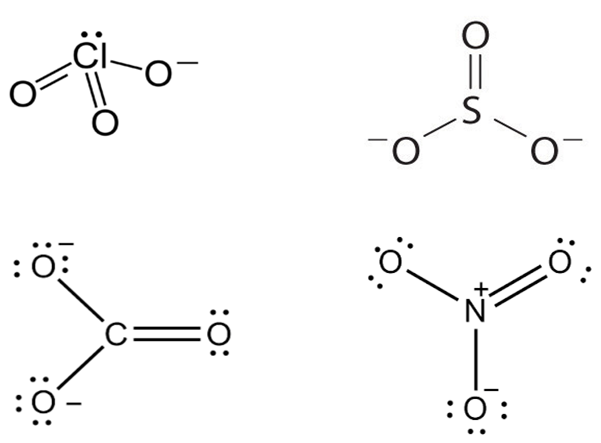
ClO3- = SO32-
Number of electrons
CO32- = 6+2+24 =32
SO32- = 16+2+24 = 32
ClO32- = 17+24+1 = 42
NO32- = 7+2+24 = 33
Hence ClO32- and SO32- are isoelectronic and are pyramidal in shape.
The number of d- electrons in Fe2+ (Z = 26) is not equal to the number of electrons in which one of the following?
S- electronic in Mg (Z=12)
p-electrons in Cl( Z=17)
d- electrons in Fe (Z=26)
d- electrons in Fe (Z=26)
which of the following orders of ionic radii is correctly represented?
H- > H >H+
Na+ >F- >O2-
F- > O2->Na+
F- > O2->Na+
Identify the wrong statement in the following.
Amongst isoelectronic species, smaller the positive charge on the cation, smaller is the ionic radius
Amongst isoelectronic species, greater the negative charge on the anion, larger is the ionic radius
Atomic radius of the elements increases as one moves down the first group of the periodic table
Atomic radius of the elements increases as one moves down the first group of the periodic table
Which of the following represents the correct order of increasing electron gain enthalpy with negative sign for the elements O, S, F and Cl?
Cl < F < O < S
O < S < F< Cl
F < S < O < Cl
F < S < O < Cl
