 Multiple Choice Questions
Multiple Choice QuestionsWhen an alpha particle of mass m moving with velocity v bombards on a heavy nucleus of charge Ze, its distance of closest approach from the nucleus depends on mass as,

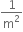
m
m
D.
m
When an alpha particle moving with velocity v bombards on a heavy nucleus of charge Ze, then there will be no loss of energy.
Initial Kinetic energy of the alpha particle = Potential energy of alpha particle at closest approach.
That is,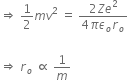
This is the required distance of closest approach to alpha particle from the nucleus.
The half-life to a radioactive nucleus is 50 days. The time interval (t2-t1) between the time t2 when 2/3 of it has decayed and the time t1 when 1/3 of it had decayed is
30 days
50 days
60 days
15 days
If the nuclear radius of27 Al is 3.6 Fermi, the approximate nuclear radius of 64Cu in Fermi is
2.4
1.2
4.8
4.8
A mixture consists of two radioactive materials A1 and A2 with half-lives of 20 s and 10 s respectively. Initially, the mixture has 40 g of A1 and 160 g of A2. The amount of the two in the mixture will become equal after
60 s
80 s
20 s
20 s
The binding energy per nucleon of  nuclei are 5.60 MeV and 7.06 MeV, respectively. In the nuclear reaction,
nuclei are 5.60 MeV and 7.06 MeV, respectively. In the nuclear reaction, 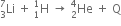 , the value of energy Q released is,
, the value of energy Q released is,
19.6 MeV
-2.4 MeV
8.4 MeV
8.4 MeV
A radio isotope X with a half life 1.4 x 109 yr decays of Y which is stable. A sample of the rock from a cave was found to contain X and Y in the ratio 1:7. The age of the rock is,
1.96 x 109 yr
3.92 x 109 yr
4.20 x 109 yr
4.20 x 109 yr
The half-life of radioactive isotope X is 20 yr. It decays to another element Y which is stable.The two elements X and Y were found to be in the ratio 1:& in a sample of given rock is estimated to be
40 yr
60 yr
80 yr
80 yr
A radioactive nucleus of mass M emits a photon of frequency v and the nucleus recoils.The recoil energy will be
h2v2 / 2Mc2
zero
hv
hv
The power obtained in a reactor using U235 disintegration is 1000kW. The mass decay of U235 per hour is
20 μg
40 μg
1 μg
1 μg
