 Multiple Choice Questions
Multiple Choice QuestionsA student takes 2 mL acetic acid in a dry test tube and adds a pinch of sodium hydrogen carbonate to it. He makes the following observations:
I. A colorless and odorless gas evolves with a brisk effervescence.
II. The gas turns lime water milky when passed through it.
III. The gas burns with an explosion when a burning splinter is brought near it.
IV. The gas extinguishes the burning splinter that is brought near it.
The correct observations are:
I, II, and III
II, III and IV
III, IV and I
III, IV and I
D.
III, IV and I
Reason: When 2 mL acetic acid is taken in a dry test tube and a pinch of sodium hydrogen carbonate is added to it, a colorless and odorless gas evolves with a brisk effervescence which is CO2. 
When CO2 is passed through lime water it turns lime water turns milky.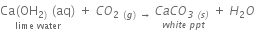
CO2 also extinguishes the burning splinter when it is brought near it.
In an experiment to study the properties of acetic acid a student takes about 2 mL of acetic acid in a dry test tube. He adds about 2 mL of water to it and shakes the test tube well. He is likely to observe that:
the acetic acid dissolves readily in water
the solution becomes light orange
water floats over the surface of acetic acid
water floats over the surface of acetic acid
A student prepared 20% sodium hydroxide solution in a beaker containing water. The observations noted by him are given below.
I. Sodium hydroxide is in the form of pellets.
II. It dissolves in water readily.
III. The beaker appears cold when touched from outside.
IV. The red litmus paper turns blue when dipped into the solution.
The correct observations are:
I, II, and III
II, III and IV
III, IV and I
III, IV and I
Read the following statements:
I. When a red litmus paper is dipped into reaction mixture of a saponification reaction, it turns blue and the reaction is exothermic.
II. When a blue litmus paper is dipped into reaction mixture of a saponification reaction, its color does not change and the reaction is exothermic.
III. When a red litmus paper is dipped into reaction mixture of a saponification reaction, its color does not change and the reaction is endothermic.
IV. When a blue litmus paper is dipped into reaction mixture of a saponification reaction, its color does not change and the reaction is endothermic.
Which of the above statements are correct?
I, and II
II and III
III and IV
III and IV
Hard water required for an experiment is not available in a school laboratory. However, following salts are available in the laboratory. Select the salts which may be dissolved in water to make it hard for the experiment.
|
(1) |
Calcium Sulphate |
(2) |
Sodium Sulphate |
|
(3) |
Calcium Chloride |
(4) |
Potassium Sulphate |
|
(5) |
Sodium Hydrogen Carbonate |
(6) |
Magnesium Chloride |
1, 2 and 4
1, 3 and 6
3, 5 and 6
3, 5 and 6
A student focused the image of a distant object using a device ‘X’ on a white screen ‘S’ as shown in the figure. If the distance of the screen from the device is 40 cm, select the correct statement about the device.
The device X is a convex lens of focal length 20 cm.
The device X is a concave mirror of focal length 40 cm.
The device X is a convex mirror of radius of curvature 40 cm.
The device X is a convex mirror of radius of curvature 40 cm.
A student obtained a sharp image of a burning candle, placed at the farther end of a laboratory table, on a screen using a concave mirror. For getting better value of focal length of the mirror, the subject teacher suggested him for focusing a well illuminated distant object. What should the student do?
He should move the mirror away from the screen.
He should move the mirror slightly towards the screen.
He should move the mirror as well as the screen towards the newly selected object.
He should move the mirror as well as the screen towards the newly selected object.
After tracing the path of rays of light through a glass slab for three different angles of incidence, a student measured the corresponding values angle of refraction r and angle of emergence e and recorded them in the table given below:
|
S. No. |
∠i |
∠r |
∠e |
|
I |
30° |
20o |
31o |
|
II |
40o |
25o |
40o |
|
III |
50o |
31o |
49o |
A)
B)
C) I and III
D) I, II and III
I and II
II and III
I and III
I and III
Select from the following the best set-up for tracing the path of a ray of light through a rectangular glass slabs:
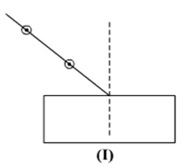
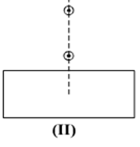
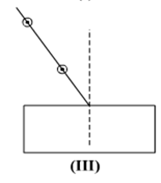
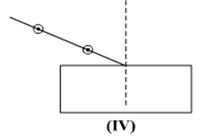
In an experiment to trace the path of a ray of light through a glass prism for different values of angle of incidence a student would find that the emergent ray:
is parallel to the incident ray
Perpendicular to the incident ray
is parallel to the refracted ray
is parallel to the refracted ray
