 Multiple Choice Questions
Multiple Choice QuestionsA student takes about 2 mL ethanoic acid in a dry test tube and adds a pinch of sodium hydrogen carbonate to it. He reports the following observations:
I. Immediately a colourless and odourless gas evolves with a brisk effervescence.
II. The gas turns lime water milky when passed through it.
III. The gas burns with an explosion when a burning splinter is brought near it.
IV. The gas extinguishes the burning splinter that is brought near it.
The correct observations are
I, II and III
II, III and IV
III, IV and I
III, IV and I
D.
III, IV and I
In an experiment to study the properties of ethanoic acid, a student takes about 3 mL of ethanoic acid in a dry test tube. He adds an equal amount of distilled water to it and shakes the test tube well. After some time he is likely to observe that
a colloid is formed in the test tube.
the ethanoic acid dissolves readily in water.
the solution becomes light orange.
the solution becomes light orange.
We need 20% aqueous solution of sodium hydroxide for the study of saponification reaction. When we open the lid of the bottle containing solid sodium hydroxide we observe it in which form?
Colourless transparent beads
Small white beads
White pellets/flakes
White pellets/flakes
While studying saponification reaction, a student measures the temperature of the reaction mixture and also finds its nature using blue/red litmus paper. On the basis of his observations the correct conclusion would be
the reaction is exothermic and the reaction mixture is acidic.
the reaction is endothermic and the reaction mixture is acidic.
the reaction is endothermic and the reaction mixture is basic.
the reaction is endothermic and the reaction mixture is basic.
In a locality, hard water, required for an experiment, is not available. However, the following salts are available in the school laboratory:
1. Sodium sulphate
2. Calcium sulphate
3. Magnesium chloride
4. Sodium chloride
5. Calcium chloride
6. Potassium sulphate
Which of the above salts may be dissolved in water to obtain hard water for the experiment?
2, 3 and 5
1, 2 and 5
1, 2, 4and 6
1, 2, 4and 6
A student focused the Sun rays using an optical device 'X' on a screen S as shown.
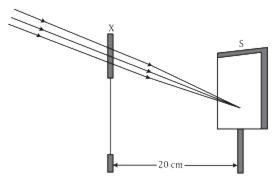
Convex lens of focal length 10 cm.
Convex lens of radius of curvature 20 cm
Convex lens of focal length 20 cm
Convex lens of focal length 20 cm
A student has obtained an image of a well-illuminated distant object on a screen to determine the focal length, F1 of the given spherical mirror. The teacher then gave him another mirror of focal length, F2 and asked him to obtain a focused image of the same object on the same screen. The student found that in order to focus the same object using the second mirror, he has to move the mirror away from the screen. From this observation, it may be concluded that both the spherical mirrors given to the student were (select the correct option):
Concave and F1 < F2
Concave and F1 > F2
Convex and F1 < F2
Convex and F1 < F2
In the following diagram, the path of a ray of light passing through a glass prism is shown:
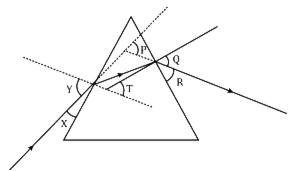
X, R and T
Y, Q and T
X,Q and P
X,Q and P
Study the following diagrams in which the path of a ray of light passing through a glass prism as traced by four students P. Q, R and S are as shown: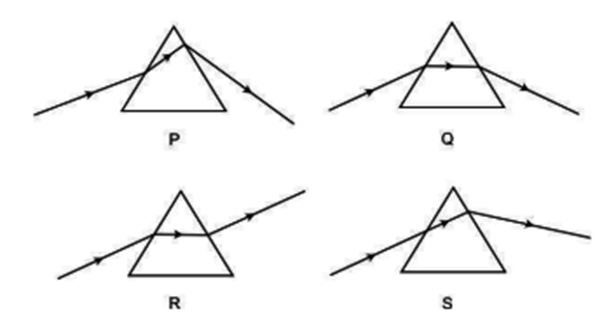
The student who has traced the path correctly is:
P
Q
R
R
A student is using a convex lens of focal length 18 cm to study the image formation by it for the various positions of the object. He observes that when he places the object at 27 cm, the location of the image is at 54 cm on the other side of the lens. Identify from the following diagram the three rays that are obeying the laws of refraction and may be used to draw the corresponding ray diagram.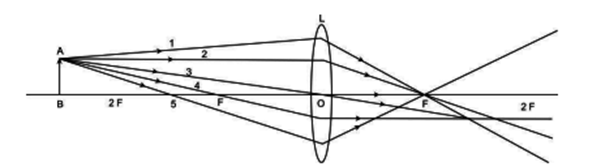
1, 2 and 4
1,3 and 5
2, 4 and 5
2, 4 and 5
