 Short Answer Type
Short Answer TypeWhat is the cause of a feeling of depression in human beings? Name a drug which can be useful in treating this depression.
Explain the following behaviours:
(i) Alcohols are more soluble in water than the hydrocarbons of comparable molecular masses.
(ii) Ortho-nitro phenol is more acidic than ortho-methoxyphenol.
(i) As alcohol is a polar solvent .it form hydrogen bonding with water molecules while another hydrocarbon of comparable molecular masses does not form hydrogen bonding due to being non- polar i.e.
Alcohols undergo intermolecular H-bonding while hydrocarbons do not.![]()
Hence, they are more soluble in water than the hydrocarbons of comparable molecular masses.
(ii) 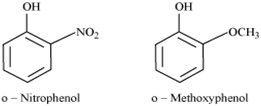
The nitro-group is an electron-withdrawing group. The presence of this group in the ortho position decreases the electron density in the O-H bond. As a result, it is easier to lose a proton.
Also, the o-nitro phenoxide ion formed after the loss of protons is stabilised by resonance.
Hence, ortho-nitrophenol is a stronger acid.
On the other hand, a methoxy group is an electron-releasing group. Thus, it increases the electron density in the O-H bond and hence, the proton cannot be given out easily.
For this reason, ortho-nitrophenol is more acidic than ortho-methoxyphenol.
Explain the mechanism of acid catalysed hydration of an alkene to form corresponding alcohol.
Complete the following chemical reaction equations:
(i) C6H5N2Cl + H3PO2+ H2O --->
(ii) C6H5NH2+ Br2(aq.)--->
Describe the following giving the relevant chemical equation in each case:
(i) Carbylamines reaction
(ii) Hofmann’s bromamide reaction.
Answer the following question:
(i) What is meant by the chirality of a compound? Give an example.
(ii) Which one of the following compounds is more easily hydrolyzed by KOH and why?
CH3CHCICH2CH3 or CH3CH2CH2Cl
(iii) Which one undergoes S N 2 substitution reaction faster and why?![]()
Define the following as related to proteins:
(i) Peptide linkage
(ii) Primary structure
(iii) Denaturation
Differentiate between thermoplastic and thermosetting polymers. Give one example of each.
 Long Answer Type
Long Answer Type(a) Draw the molecular structure of the following compounds.
(i) N2O5
(ii) XeOF4
(b) Explain the following observation:
(i) Sulphur has a greater tendency for catenation than oxygen.
(ii) ICI is more reactive than I2.
(iii) Despite the lower value of its electron gain enthalpy with a negative sign, fluorine (F2) is a stronger oxidizing agent than Cl2.
