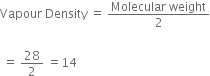Short Answer Type
Short Answer TypeGive the correct IUPAC name and the functional group for each of the compounds whose structural formulae are given below:
Give the correct IUPAC name and the functional group for each of the compounds whose structural formulae are given below: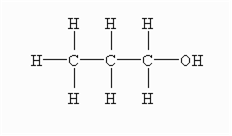
Draw the structural formulae of the two isomers of Butane. Give the correct IUPAC name of each isomer.
 Long Answer Type
Long Answer Typei) Calculate the number of moles and the number of molecules present in 1.4g of ethylene gas . what is the volume occupied by the same amount of ethylene?
ii) What is the vapour density of ethylene?
(Avogadro's number =6 x1023; Atomic weight of C=12, H=1; Molar volume =22.4litres at STP)
Molecular weight of ethylene =CH2=CH2
= 12+2+12+2 =28g
Number of moles = 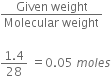
Number of molecules in 1 mole = 6 x 1023
Therefore,
Number of molecules in 0.05 mole =6x1023x0.05 =0.3 x 1023
=3 x 1022 molecules.
Volume of occupied by 1 mole = 22.4l
volume of occupied by 0.05 = 22.4 x 0.05 = 1.12 litre
ii)