 Multiple Choice Questions
Multiple Choice QuestionsFor a spontaneous reaction the ∆G, equilibrium constant (K) and Eocell will be respectively
-ve, >1, +ve
+ve, >1, -ve
-ve, <1, -ve
-ve, <1, -ve
Consider the reaction: N2 +3H2 → 2NH3 carried out at constant temperature and pressure. If ∆H and ∆U are the enthalpy and internal energy changes for the reaction, which of the following expressions is true?
∆H = 0
∆H = ∆U
∆H < ∆U
∆H < ∆U
Which one of the following statements is NOT true about the effect of an increase in temperature on the distribution of molecular speeds in a gas?
The most probable speed increases
The fraction of the molecules with the most probable speed increases
The distribution becomes broader
The distribution becomes broader
The exothermic formation of ClF3 is represented by the equation
Which of the following will increase the quantity of ClF3 in an equilibrium mixture of Cl2, F2 and ClF3?
Increasing the temperature
Removing Cl2
Increasing the volume of the container
Increasing the volume of the container
For the reaction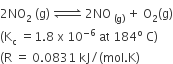
When Kp and Kc are compared at 184oC, it is found that
Kp is greater than Kc
Kp is less than Kc
Kp = Kc
Kp = Kc
Hydrogen ion concentration in mol / L in a solution of pH = 5.4 will be
3.98 x 108
3.88 x 106
3.68 x 10-6
3.68 x 10-6
If we consider that 1/6 in place of 1/12 mass of carbon atom is taken to be the relative atomic mass unit, the mass of one mole of a substance will
Decrease twice
Increase two fold
Remain unchanged
Remain unchanged
In a multi-electron atom, which of the following orbitals described by the three quantum numbers will have the same energy in the absence of magnetic acid and electric fields?
(a) n = 1, l = 0, m = 0
(b) n = 2, l = 0, m = 0
(c) n = 2, l = 1, m = 1
(d) n = 3, l = 2, m = 1
(e) n = 3, l = 2, m = 0
(a) and (b)
(b) and (c)
(c) and (d)
(c) and (d)
The number and type of bonds between two carbon atoms in calcium carbide are
One sigma, one pi
One sigma, two pi
Two sigma, one pi
Two sigma, one pi