 Multiple Choice Questions
Multiple Choice QuestionsThe molar conductivities of KCl, NaCl and KNO3 are 152, 128 and 111 S cm2mol-1 respectively. What is the molar conductivity of NaNO3?
101 S cm2mol-1
87 S cm2mol-1
-101 S cm2mol-1
-39 S cm2mol-1
The electrochemical cell stops working after sometime because
electrode potential of both the electrodes becomes zero
electrode potential of both the electrodes becomes equal
one of the electrodes is eaten away
the cell reaction gets reversed
The amount of electricity required to produce one mole of copper from copper sulphate solution will be
1 F
2.33 F
2 F
1.33 F
Dipping iron article into a strongly alkaline solution of sodium phosphate
does not affect the article
forms iron phosphate film
forms ferric hydroxide
When acetamide is hydrolysed by boiling with acid, the product obtained is
acetic acid
ethyl amine
ethanol
acetamide
A.
acetic acid
In presence of dil HCl, acetamide is hydrolysed by boiling, the product obtained is acetic acid (CH3COOH).
Secondary nitroalkanes can be converted into ketones by using Y. Identify Y from the following
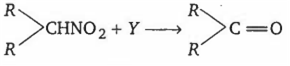
aqueous HCl
aqueous NaOH
KMnO4
CO
Alkyl cyanides undergo Stephen reduction to produce
aldehyde
secondary amine
primary amine
amide
Which of the following set consists only of essential amino adds?
Alanine, tyrosine, cystine
Leucine, lysine, tryptophane
Alanine, glutamine, lycine
leucine, praline, glycine
