 Multiple Choice Questions
Multiple Choice QuestionsWhen 0.2 g of 1-butanol was burnt in a suitable apparatus, the heat evolved was sufficient to raise the temperature of 200 g water by 5°C. The enthalpy of combustion of 1-butanol in kcal mol-1 will be
+37
+370
-370
-740
Given that dE = Tds - pdV and H = E + pV. Which one of the following relations is true ?
dH = TdS + Vdp
dH = SdT + Vdp
dH = -SdT + Vdp
dH = dE + pdV
When 200 mL of aqueous solution of HCl (pH = 2) is mixed with 300 mL of an aqueous solution of NaOH (pH = 12) the pH of the resulting mixture is
10
2.7
4.0
11.3
At a certain temperature, the dissociation constants of formic acid and acetic acid are 1.8 × 10-4 and 1.8 × 10-5 respectively. The concentration of acetic acid solution in which the hydrogen ion has the same concentration as in 0.001 M formic acid solution is equal to
0.01 M
0.001 M
0.1 M
0.0001 M
In acid medium, Zn reduces nitrate ion to NH ion according to the reaction,
Zn + NO→ Zn2+ + NH + H2O (unbalanced).
How many moles of HCl are required to reduce half a mole of NaNO3 completely ? Assume the availability of sufficient Zn.
5
4
3
2
Match List-I and List-II and choose the correct matching codes.
| List - I | List - II |
| A. [Ni(CN)4]2- | 1. Ti4+ |
| B. CHlorophyll | 2. sp3; paramagentic |
| C. Ziegler- Natta catalyst | 3. non-planar |
| D. [NiCl4]2- | 4. Mg2+ |
| E. Deoxyhaemoglobin |
5. Planar 6. dsp2; diamagentic |
A - 6; B - 4; C - 1; D - 2; E - 3
A - 2; B - 4; C - 1; D - 6; E - 3
A - 2; B - 4; C - 1; D - 6; E - 5
A - 6; B - 4; C - 1; D - 2; E - 5
A.
A - 6; B - 4; C - 1; D - 2; E - 3
Among the given options, option a is the correct one.
a. [Ni(CN)4]2-
Ni(28) = [Ar] 3d8 4s2
Ni2+ = [Ar] 3d8, 4s
Since, CN- is a strong field ligand, it causes pairing of electrons.
[Ni(CN)4]2- =

b. Chlorophyll consists of Mg2+ ion.
c. Zeigler-Natta catalyst is Ti4+ + (C2H5)3Al.
d. [NiCl4]2-
In this case, Cl- is a weak field ligand, so does not pairing. Hence,
[NiCl4]2- =
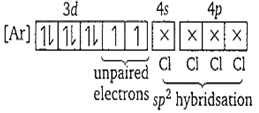
Due to the presence of unpaired electrons, it is paramagentic.
e. Deoxyhaemoglobin is non-planar while oxyhaemoglobin is planar.
What is the overall formation equilibriumconstant for the ion [ML4]2- ion, given that β4 for this complex is 2.5 × 1013?
2.5 × 1013
5 × 10-13
2.5 × 10-14
4.0 × 10-13
In which of the following species, all the three types of hybrid carbons are present ?
CH2=C=CH2
CH3-CH=CH-CH
CH3-C≡C-CH
CH3-CH=CH-CH
Which one of the following pairs of substances will not produce hydrogen when reacted together?
Copper and conc. nitric acid
Ethanol and metallic sodium
Magnesium and steam
Phenol and metallic sodium
A compound in which a metal ion Mx+ (Z = 25) has a spin only magnetic moment of BM. The number of unpaired electrons in the compound and the oxidation state of the metal ion are respectively
4 and 2
5 and 3
3 and 2
4 and 3
