 Multiple Choice Questions
Multiple Choice QuestionsConsider the following reaction,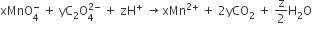
The values of x,y and z in the reaction are respectively
5,2 and 16
2,5 and 8
2,5 and 16
2,5 and 16
The gas leaked from a storage tank of the Union Carbide plant in Bhopal tragedy was
Methyl isocyanate
Methylamine
Ammonia
Ammonia
Which of the following complex species is not expected to exhibit optical isomerism?
[Co(en)3]3+
[Co(en)2Cl2]+
[Co(NH3)3Cl3]
[Co(NH3)3Cl3]
C.
[Co(NH3)3Cl3]
Complexes of the type [MA2(AA2), [M(AA3)] exhibit optical isomerism.
Optical Isomerism is shown by only those complexes which lack elements of symmetry.
In the given complexes, [Co(NH3)3Cl3] shows facial as well as meridional isomerism. But both of the forms contain a plane of symmetry. Thus, only [Co(NH3)3Cl3] complex does not show optical isomerism.
Which one of the following molecules is expected to exhibit diamagnetic behaviour?
C2
O2
N2
N2
Given, 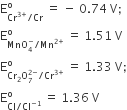
Based on the data given above, strongest oxidising agent will be
Cl
Cr3+
Mn2+
Mn2+
The molarity of a solution obtained by mixing 750 mL of 0.5 (M) HCl with 250 mL of 2(M) HCl will be
0.875 M
1.00 M
1.75 M
1.75 M
The rate of a reaction doubles when its temperature changes from 300K to 310K. Activation energy of such a reaction will be (R = 8.314 JK–1 mol–1 and log 2 = 0.301)
53.6 kJ mol-1
48.6 kJ mol-1
58.5 kJ mol-1
58.5 kJ mol-1
Which of the following arrangements does not represent the correct order of the property stated against it?
V2+ < Cr2+<Mn2+<Fe2+: paramagnetic behaviour
Ni2+ < Co2+ < Fe2+ < Mn2+ ; Ionic size
Co3+ < Fe3+< Cr3+ < Sc3+ : stability in aqueous solution
Co3+ < Fe3+< Cr3+ < Sc3+ : stability in aqueous solution
Which of the following is the wrong statement?
ONCl and ONO- are not isoelectronic
O3 molecule is linear
Ozone is violet-black in solid state
Ozone is violet-black in solid state
The coagulating power of electrolytes having ions Na+, Al3+ and Ba2+ for arsenic sulphide sol increases in the order:
Al3+<Ba2+<Na+
Na+<Ba2+<Al3+
Ba2+< Na2+<Al3+
Ba2+< Na2+<Al3+
