 Multiple Choice Questions
Multiple Choice QuestionsSelect the correct statement(s).
LiAlH4 reduces methyl cyanide to methyl amine.
Alkaline nitrile has electrophilic as well as nucleophilic centres
Saponification is a reversible reaction.
Alkaline hydrolysis of methane nitrile forms methanoic acids.
In a Claisen condensation reaction (when an ester is treated with a strong base)
a proton is removed from the α-carbon to form a resonance stabilised carbanion of the ester
carbanion acts as a nucleophile in a nucelophilic acyl substitution reaction with another ester molecule
a new C - C bond is formed
All of the above statements are correct
An organic compound B is formed by the reaction of ethyl magnesium iodide with a substance A, followed by treatment with dilute aqueous acid. Compound B does not react with PCC or PDC in dichloromethane. Which of the following is a possible compound for A?
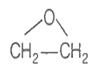
CH3CH2COCH3
CH3COH
H2C=O
Benzene diazonium chloride on treatment with hypophosphorus acid and water yield benzene. Which of the following is used as a catalyst in this reaction?
LiAlH4
Red P
Zn
Cu+
Consider the following reaction sequence,
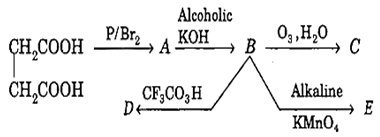
Isomers are
C and E
C and D
D and E
C, D and E
C.
D and E
Isomers in the above given reaction are D and E. The reaction can be written as follows-

When a monosaccharide forms a cyclic hemiacetal, the carbon atom that contained the carbonyl group is identified as the ....... carbon atom, because
D, the carbonyl group is drawn to the right
L, the carbonyl group is drawn to the left
acetal, it forms bond to an -OR and an -OR'
anomeric, its substitutents can assume an α or β position
