 Multiple Choice Questions
Multiple Choice QuestionsSelect the coloured compound amongst the following .
(At. no. Ti= 22, Cr= 24, Cu= 29, Zn= 30)
TiCl4
CrCl3
ZnCl3
CuCl
Which among the following solids crystallises as a face centred cube?
Iron
Rubidium
Uranium
Platinum
Which oxyacid of sulphur contains S-S single bond?
Oleum
Marshall's acid
Dithionic acid
Thiosulphuric acid
Select the diamagnetic complex ion amongst the following complexes (At. no. : Fe = 26,CO = 27)
K3[Fe(CN)6]
[Co(NH3)6)Cl3
K3[FeF6]
K3[CoF6]
Write IUPAC name of following compound
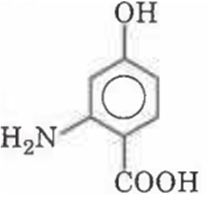
2-amino-4-hydroxybenzoic acid
6-amino-4-hydroxybenzoic acid
3-amino-4-carboxyphenol
2-carboxy-4-hydroxyaniline
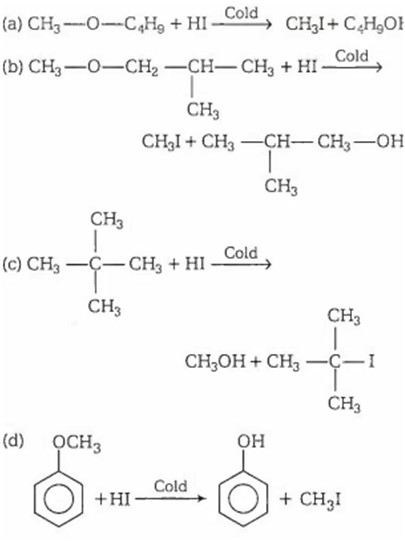
Select the ether among following that yields methanol as one the products on reaction with cold hydroiodic acid
1-methoxybutane
1-methoxybutane-2-methylpropane
2-methoxy-2-methylpropane
methoxybenzene
C.
2-methoxy-2-methylpropane
According Zeise! method, when an ether reacts with cold HI, alkyl iodide and alcohol are formed. In case of asymmetrical ether, the alkyl halide is always formed from the smaller alkyl group provided no tertiary (3°) alkyl group is present and if any 3° alkyl group is present, the halogen gets attached with it. In case of alkyl aryl ethers, the products are always phenol and an alkyl halide.
The two monomers used in the preparation of dextron are
3-hydroxy butanoic acid and 3-hydroxy pentanoic acid
∈ amino caproic acid and glycine
isobutylene and isoprene
lactic acid and glycolic acid
One mole of stachyose on hydrolysis yields
1 mole of glucose+ 1 mole of fructose+ 2 mole of galactose
2 mole of glucose + 1 mole of fructose + 1 mole of galactose
1 mole of glucose+ 2 mole of fructose+ 1 mole of galactose
2 mole of glucose+ 2 mole of fructose
Alkaline hydrolysis of which among the following compounds leads to the formation of a racemate?
1-bromo-1-phenylethane
1-chloro-3-methylbutane
Bromoethane
1-chloropropane
