 Multiple Choice Questions
Multiple Choice QuestionsDipole-dipole interaction energy between polar molecules in solids depends on the radius of the molecule (r) and it is directly proportional to
(1/r3)
(1/r6)
(1/r)
(1/r5)
Critical density of a gas having molecular weight 39 g mol-1 is 0.1 × 103 g cm-3. Its critical volume in L mol-1 is
0.390
3.90
0.039
39.0
The low solubility of LiF and that of CsI in water are respectively due to which of the following properties of the alkali metal ions?
Higher hydration enthalpy of Li, higher lattice enthalpy of Cs+
Smaller hydration enthalpy of Li, higher lattice enthalpy of Cs+
Smaller lattice enthalpy of Li, higher hydration enthalpy of Cs+
Higher lattice enthalpy of Li, smaller hydration enthalpy of Cs+
In aqueous solution, Cr2+ is a stronger reducing agent than Fe2+. This is because
Cr2+ ion is more stable than Fe2+
Cr3+ ion with d3 configuration has favourable crystal field stabilisation energy
Cr3+ has half filled configuration and hence more stable
Fe3+ in aqueous solution is more stable than Cr3+
The major product obtained by the addition reaction of HBr to 4-methylpent-1-ene in the presence of peroxide is
1-bromo-4-methylpentane
4-bromo-2-methylpentane
2-bromo-4-methylpentane
3-bromo-2-methylpentane
Some organic compounds are given in List I and their uses in List II. Choose the correct matching.
| List I | List II |
| A. Triiodomethane | i. Solvent for alkaloids |
| B. p, p'-dichlorodiphenyl-trichloroethane | ii. Propellent in aerosols |
| C. Trichloromethane | iii. Antiseptic |
| D. Dichloromethane | iv. Insecticide |
A - ii; B - iv; C - i; D - iii
A - iii; B - iv; C - i; D - ii
A - ii; B - i; C - iv; D - iii
A - i; B - iii; C - ii; D - iv
The total number of monohalogenated products formed by halogenation of 2, 4, 4-trimethylhexane are,
5
7
6
8
C.
6
Total number of monohalogenated products of 2,4,4-trimethylhexane are six.
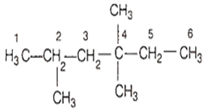
(i) 2 monohalogenated products by replacing two CH3 groups at C2 and C4 position.
(ii) 4 monohalogenated products by replacing H by Cl at C2, C3, C5, C6 positions. Thus, total 6 isomers are possible.
