 Multiple Choice Questions
Multiple Choice QuestionsWhen potassium is reacted with water which compound(s) is are formed preferentially?
K2O
KO2
Both K2O and KO2
K2O2
Select the most appropriate statement In BF3.
All the bonds are completely ionic.
All the bonds are covalent.
The B-F bond is partially ionic.
B-F bond has partial double bond character.
When MnO2 is fused with KOH and KNO2 , a coloured compound is formed. Choose the right compound with the appropriate colour.
K2MnO4, green
KMnO4, purple
Mn2O3, brown
Mn3O4, black
Identify the case(s) where there is change is oxidation number
Acidified solution of CrO
SO2 gas bubbled through an acidic solution Cr2O
Alkaline solution of CrO
Ammoniacal solution of CrO
The solution which does not produce precipitate when treated with aqueous K2CO3 is
BaCl2
CaBr2
MgCl2
Na2SO4
Lassaigne's test (with silver nitrate) is commonly used to detect halogens such as chlorine, bromine and iodine but not useful to detect fluorine because the product AgF formed is
Volatile
Reactive
Explosive
Soluble in water
The reaction in propene with HBr is presence of peroxide proceeds through the intermediate
H3C - H - CH3
H3C - H -CH2Br
H3C - H2
H3C - CH2 - H2
B.
H3C - H -CH2Br
The reaction in propene with HBr in presence of peroxide proceeds through the intermediate H3C - H -CH2Br. The intermediate produce in case of addition of HBr occurs according to anti-markonicov's rule.
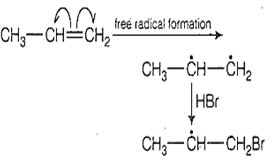
Cycloheptatrienyl cation is
Non-benzenoid and non-aromatic
Non-benzenoid and aromatic
Benzenoid and non-aromatic
Benzenoid and aromatic
Which of the following is not true about a catalyst?
Mechanism of the reaction in presence and absence of catalyst could be different.
Use of catalyst cannot change the order of the reaction
Catalyst enhances both forward and backwards reaction at equal rate.
Catalyst participates in the reaction, but not consumed in the process.
