 Multiple Choice Questions
Multiple Choice QuestionsThe osmotic pressure of a dilute solution of an ionic compound XY in water is four times that of a solution of 0.01 M BaCl2 in water. Assuming complete dissociation of the given ionic compounds is water, the concentration of XY (in mol L-1 ) in solution is
4 × 10-4
16 × 10-4
4 × 10-2
6 × 10-2
Aniline dissolved in dilute HCl is reacted with sodium nitrite at 0ºC. This solution was added dropwise to a solution containing equimolar mixture of aniline and phenol in dil. HCl. The structure of the major product is
![]()
![]()
![]()
![]()
The major product of the following reaction is
CH3C≡CH
CH3C (I) Cl CHD2
CH3CD (Cl) CHD (I)
CH3CD2CH(Cl) (I)
CH3CD(I)CHD (Cl)
A.
CH3C (I) Cl CHD2
The major product is
The increasing order of reactivity of the following compounds towards aromatic electrophilic substitution reaction is
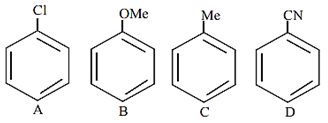
D < A < C < B
B < C < A < D
D < B < A< C
A < B < C < D
Which of the following statement is not true about sucrose?
On hydrolysis, it produces glucose and fructose
It is also named as invert sugar
It is non-reducing sugar
The glycosidic linkage is present between C1 of α-glucose and C1 of β- fructose.
The major product of the following reaction is
CH3CH=CHCO2CH3
CH3CH2CH2CO2CH3
CH3CH2CH2CH2OH
CH3CH2CH2CHO
CH3CH=CHCH2OH
