 Multiple Choice Questions
Multiple Choice QuestionsThe electronic configuration is 1s2 2s2 2p5 3s1 shows
ground state of fluorine atom
excited state of fluorine atom
excited state of neon atom
excited state of ion O2
Assertion: Relative strength of acids can be known by knowing the value of dissociation constant.
Reason : It gives the value of H dissolved in solution.
If both Assertion and Reason are true and Reason is the correct explanation of Assertion.
If both Assertion and Reason are true but Reason is not the correct explanation of Assertion.
If Assertion is true but Reason is false.
If Assertion is false but Reason is true.
Assertion : CHCl3 and CH3OH are miscible.
Reason : One of them is polar.
If both Assertion and Reason are true and Reason is the correct explanation of Assertion
If both Assertion and Reason are true but Reason is not the correct explanation of Assertion.
If Assertion is true but Reason is false.
If Assertion is false but Reason is true.
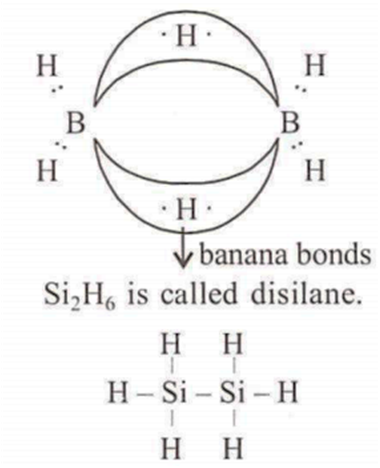
Assertion : B2H6, Si2H6, are said to have similar structure.
Reason : They have same number of and bonds.
If both Assertion and Reason are true and Reason is the correct explanation of Assertion.
If both Assertion and Reason are true but Reason is not the correct explanation of Assertion.
If Assertion is true but Reason is false.
If Assertion is false but Reason is true.
D.
If Assertion is false but Reason is true.
B2H6 is an electron deficient compound. B2H6 contain some unusual bonds which are called as 2-electron 3-centre bonds.
Assertion : Hydrogen nuclei combines to form helium nuclei then energy is released.
Reason: Binding energy/nucleon of He is greater than hydrogen.
If both Assertion and Reason are true and Reason is the correct explanation of Assertion
If both Assertion and Reason are true but Reason is not the correct explanation of Assertion.
If Assertion is true but Reason is false.
If Assertion is false but Reason is true.
Assertion : Water is used as a moderator in nuclear reactor.
Reason : Moderator is a light substance that absorb neutrons.
If both Assertion and Reason are true and Reason is the correct explanation of Assertion.
If both Assertion and Reason are true but Reason is not the correct explanation of Assertion.
If Assertion is true but Reason is false.
If Assertion is false but Reason is true.
Assertion : Ionisation potential of B (atomic no.5) is less than Be (atomic no. 4).
Reason : The first electron released from Be is of p-orbital but that from B is of s-orbital.
If both Assertion and Reason are true and Reason is the correct explanation of Assertion.
If both Assertion and Reason are true but Reason is not the correct explanation of Assertion.
If Assertion is true but Reason is false.
If Assertion is false but Reason is true.
