 Multiple Choice Questions
Multiple Choice QuestionsAn element 'Y' emits one -and two - particles to give 'X'. X and Y are
isotones
isobars
isotopes
isoelectronics
C.
isotopes
Let atomic mass and atomic number of Y be 'a' and 'b' respectively. Hence,
bYa b-2Ma-4 b-1Na-4 bXa-4
Hence, X and Y are isotopes i.e., they have equal atomic number but different in mass number.
For the reaction, 2NO2 2NO + O2, the value of Kp is equal to K. What will the value of Kp for reaction,
NO + O2 NO2
Which one of the following elements, is a component of a compound essential for photosynthesis ?
Iron
Calcium
Barium
Magnesium
The correct order of stability of carbocations, is :
1 < 2 < 3 < CH3
H3 < 1 < 2 < 3
3 < 2 < 1 < H3
2 < 3 < 1 < H3
The IUPAC name of the following compound is
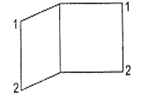
bicyclo [2, 2, 0] octane
bicyclo [0, 2, 2] hexane
bicyclo [2, 1, 1] hexane
bicyclo [2, 2, 0] hexane
The following compound can not exhibit
CH3CH = CH - CH(OH).COOH
geometrical isomerism
optical isomerism
metamerism
position isomerism
