 Multiple Choice Questions
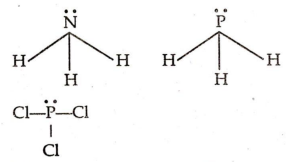
Multiple Choice QuestionsWhich of the following does not have a lone pair on the central atom ?
NH3
PH3
BF3
PCl3
C.
BF3
BF3 is a electron deficient compound .So it has no lone pair orbital over B atom .
What is the nature of the bond between B and O in (C2H5)2OBH3 ?
Covalent
Co - ordinate covalent
Ionic bond
Banana shaped bond
Following pair is separated by yellow ammonium sulphide :
CdS , Bi2S1
Bi2S3 , PbS
PbS , HgS
CdS , Ag2S3
